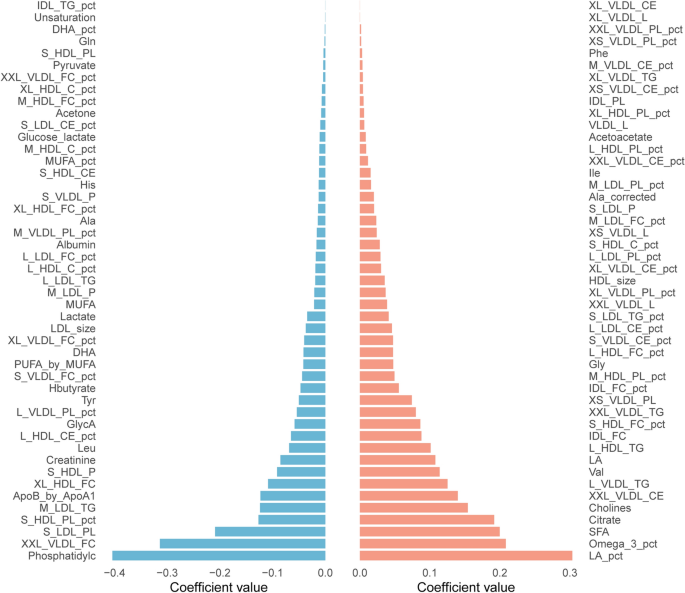
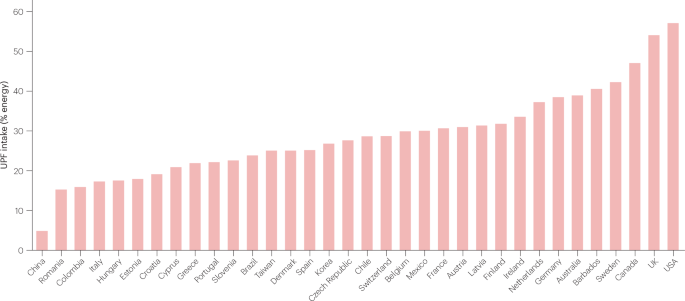
The role of ultra-processed food in obesity
World Obesity Federation. World obesity atlas 2023. World Obesity www.worldobesity.org/resources/resource-library/world-obesity-atlas-2023 (2023).
Swinburn, B. A. et al. The global obesity pandemic: shaped by global drivers and local environments. Lancet 378, 804–814 (2011).
Google Scholar
Swinburn, B. A. et al. The global syndemic of obesity, undernutrition, and climate change: the Lancet Commission Report. Lancet 393, 791–846 (2019).
Google Scholar
Popkin, B. M. Global nutrition dynamics: the world is shifting rapidly toward a diet linked with noncommunicable diseases. Am. J. Clin. Nutr. 84, 289–298 (2006).
Google Scholar
Popkin, B. M. & Gordon-Larsen, P. The nutrition transition: worldwide obesity dynamics and their determinants. Int. J. Obes. Relat. Metab. Disord. 28, S2–S9 (2004).
Google Scholar
Monteiro, C. A. Nutrition and health. The issue is not food, nor nutrients, so much as processing. Public Health Nutr. 12, 729–731 (2009).
Google Scholar
Monteiro, C. A. et al. Household availability of ultra-processed foods and obesity in nineteen European countries. Public Health Nutr. 21, 18–26 (2018).
Google Scholar
Moodie, R. et al. Profits and pandemics: prevention of harmful effects of tobacco, alcohol, and ultra-processed food and drink industries. Lancet 381, 670–679 (2013).
Google Scholar
Pereira, M. A. et al. Fast-food habits, weight gain, and insulin resistance (the CARDIA study): 15-year prospective analysis. Lancet 365, 36–42 (2005).
Google Scholar
Malik, V. S., Schulze, M. B. & Hu, F. B. Intake of sugar-sweetened beverages and weight gain: a systematic review. Am. J. Clin. Nutr. 84, 274–288 (2006).
Google Scholar
Drewnowski, A. The real contribution of added sugars and fats to obesity. Epidemiol. Rev. 29, 160–171 (2007).
Google Scholar
Lane, M. M. et al. Ultra-processed food exposure and adverse health outcomes: umbrella review of epidemiological meta-analyses. BMJ 384, e077310 (2024).
Google Scholar
Juul, F., Vaidean, G. & Parekh, N. Ultra-processed foods and cardiovascular diseases: potential mechanisms of action. Adv. Nutr 12, 1673–1680 (2021).
Google Scholar
Mialon, M., Serodio, P. & Scagliusi, F. B. Criticism of the NOVA classification: who are the protagonists? World Nutr. 9, 176–240 (2018).
NCD Risk Factor Collaboration (NCD-RisC) Worldwide trends in underweight and obesity from 1990 to 2022: a pooled analysis of 3663 population-representative studies with 222 million children, adolescents, and adults. Lancet 403, 1027–1050 (2024).
Global Network Against Food Crises, Food Security Information Network. Global report on food crises 2024. FSIN (2024).
Valicente, V. M. et al. Ultraprocessed foods and obesity risk: a critical review of reported mechanisms. Adv. Nutr. 14, 718–738 (2023).
Google Scholar
Monda, A. et al. Ultra-processed food intake and increased risk of obesity: a narrative review. Foods 13, 2627 (2024).
Google Scholar
Shim, J. S. Ultra-processed food consumption and obesity: a narrative review of their association and potential mechanisms. J. Obes. Metab. Syndr. 34, 27–40 (2025).
Google Scholar
de Araujo, T. P., de Moraes, M. M., Afonso, C., Santos, C. & Rodrigues, S. S. P. Food processing: comparison of different food classification systems. Nutrients 14, 729 (2022).
Google Scholar
Scrinis, G. & Monteiro, C. From ultra-processed foods to ultra-processed dietary patterns. Nat. Food 3, 671–673 (2022).
Google Scholar
Aguilera, J. M. The food matrix: implications in processing, nutrition and health. Crit. Rev. Food Sci. Nutr. 59, 3612–3629 (2019).
Google Scholar
Weaver, C. M. et al. Processed foods: contributions to nutrition. Am. J. Clin. Nutr. 99, 1525–1542 (2014).
Google Scholar
Monteiro, C. A., Moubarac, J. C., Cannon, G., Ng, S. W. & Popkin, B. Ultra-processed products are becoming dominant in the global food system. Obes. Rev. 14, 21–28 (2013).
Google Scholar
Wood, B., Williams, O., Baker, P. & Sacks, G. Behind the ‘creative destruction’ of human diets: an analysis of the structure and market dynamics of the ultra-processed food manufacturing industry and implications for human health. J. Agrarian Change 23, 811–843 (2023).
Gilmore, A. B. et al. Defining and conceptualising the commercial determinants of health. Lancet 401, 1194–1213 (2023).
Google Scholar
Fazzino, T. L., Jun, D., Chollet-Hinton, L. & Bjorlie, K. US tobacco companies selectively disseminated hyper-palatable foods into the US food system: empirical evidence and current implications. Addiction 119, 62–71 (2024).
Google Scholar
Martinez-Steele, E. et al. Best practices for applying the Nova food classification system. Nat. Food 4, 445–448 (2023).
Google Scholar
Steele, E. M. et al. Identifying and estimating ultraprocessed food intake in the US NHANES according to the Nova classification system of food processing. J. Nutr. 153, 225–241 (2023).
Google Scholar
Fangupo, L. J. et al. Relative validity and reproducibility of a food frequency questionnaire to assess energy intake from minimally processed and ultra-processed foods in young children. Nutrients 11, 1290 (2019).
Google Scholar
Oviedo-Solis, C. I. et al. A semi-quantitative food frequency questionnaire has relative validity to identify groups of NOVA food classification system among Mexican adults. Front. Nutr. 9, 737432 (2022).
Google Scholar
Oviedo-Solis, C. I., Monterrubio-Flores, E. A., Cediel, G., Denova-Gutierrez, E. & Barquera, S. Relative validity of a semi-quantitative food frequency questionnaire to estimate dietary intake according to the NOVA classification in Mexican children and adolescents. J. Acad. Nutr. Diet. 122, 1129–1140 (2022).
Google Scholar
Jung, S., Park, S. & Kim, J. Y. Comparison of dietary share of ultra-processed foods assessed with a FFQ against a 24-h dietary recall in adults: results from KNHANES 2016. Public Health Nutr. 25, 1166–1175 (2022).
Huybrechts, I. et al. Characterization of the degree of food processing in the European Prospective Investigation into Cancer and Nutrition: application of the Nova classification and validation using selected biomarkers of food processing. Front. Nutr. 9, 1035580 (2022).
Google Scholar
Sneed, N. M. et al. Reliability and validity of assigning ultraprocessed food categories to 24-h dietary recall data. Am. J. Clin. Nutr. 117, 182–190 (2023).
Google Scholar
Dinu, M. et al. Reproducibility and validity of a food-frequency questionnaire (NFFQ) to assess food consumption based on the NOVA classification in adults. Int. J. Food Sci. Nutr. 72, 861–869 (2021).
Google Scholar
Sarbagili-Shabat, C. et al. Development and validation of processed foods questionnaire (PFQ) in adult inflammatory bowel diseases patients. Eur. J. Clin. Nutr. 74, 1653–1660 (2020).
Google Scholar
Neri, D. et al. A novel web-based 24-h dietary recall tool in line with the Nova food processing classification: description and evaluation. Public Health Nutr. 26, 1997–2004 (2023).
Google Scholar
O’Connor, L. E. et al. Metabolomic profiling of an ultraprocessed dietary pattern in a domiciled randomized controlled crossover feeding trial. J. Nutr. 153, 2181–2192 (2023).
Google Scholar
Menichetti, G., Ravandi, B., Mozaffarian, D. & Barabasi, A. L. Machine learning prediction of the degree of food processing. Nat. Commun. 14, 2312 (2023).
Google Scholar
Vandevijvere, S. et al. Global trends in ultraprocessed food and drink product sales and their association with adult body mass index trajectories. Obes. Rev. 20, 10–19 (2019).
Google Scholar
Juul, F. & Hemmingsson, E. Trends in consumption of ultra-processed foods and obesity in Sweden between 1960 and 2010. Public Health Nutr. 18, 3096–3107 (2015).
Google Scholar
Marrón-Ponce, J. A., Tolentino-Mayo, L., Hernández, F. M. & Batis, C. Trends in ultra-processed food purchases from 1984 to 2016 in Mexican households. Nutrients 11, 45 (2018).
Google Scholar
Baker, P. et al. Ultra-processed foods and the nutrition transition: global, regional and national trends, food systems transformations and political economy drivers. Obes. Rev. 21, e13126 (2020).
Google Scholar
Dicken, S. J., Qamar, S. & Batterham, R. L. Who consumes ultra-processed food? A systematic review of sociodemographic determinants of ultra-processed food consumption from nationally representative samples. Nutr. Res. Rev. 37, 416–456 (2023).
Google Scholar
Mertens, E., Colizzi, C. & Penalvo, J. L. Ultra-processed food consumption in adults across Europe. Eur. J. Nutr. 61, 1521–1539 (2022).
Google Scholar
Li, M. & Shi, Z. Association between ultra-processed food consumption and diabetes in Chinese adults – results from the China Health and Nutrition Survey. Nutrients 14, 4241 (2022).
Google Scholar
Martini, D., Godos, J., Bonaccio, M., Vitaglione, P. & Grosso, G. Ultra-processed foods and nutritional dietary profile: a meta-analysis of nationally representative samples. Nutrients 13, 3390 (2021).
Google Scholar
Moradi, S. et al. Ultra-processed food consumption and adult obesity risk: a systematic review and dose-response meta-analysis. Crit. Rev. Food Sci. Nutr. 63, 249–260 (2023).
Google Scholar
Askari, M., Heshmati, J., Shahinfar, H., Tripathi, N. & Daneshzad, E. Ultra-processed food and the risk of overweight and obesity: a systematic review and meta-analysis of observational studies. Int. J. Obes. 44, 2080–2091 (2020).
Lane, M. M. et al. Ultraprocessed food and chronic noncommunicable diseases: a systematic review and meta-analysis of 43 observational studies. Obes. Rev. 22, e13146 (2021).
Google Scholar
Pagliai, G. et al. Consumption of ultra-processed foods and health status: a systematic review and meta-analysis. Br. J. Nutr. 125, 308–318 (2021).
Google Scholar
Bestari, F. F., Andarwulan, N. & Palupi, E. Synthesis of effect sizes on dose response from ultra-processed food consumption against various noncommunicable diseases. Foods 12, 4457 (2023).
Google Scholar
Guyatt, G. et al. GRADE guidelines: 1. Introduction – GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 64, 383–394 (2011).
Google Scholar
Konieczna, J. et al. Contribution of ultra-processed foods in visceral fat deposition and other adiposity indicators: prospective analysis nested in the PREDIMED-Plus trial. Clin. Nutr. 40, 4290–4300 (2021).
Google Scholar
Li, M. & Shi, Z. Ultra-processed food consumption associated with overweight/obesity among Chinese adults – results from China Health and Nnutrition Survey 1997-2011. Nutrients 13, 2796 (2021).
Google Scholar
Cordova, R. et al. Consumption of ultra-processed foods associated with weight gain and obesity in adults: a multi-national cohort study. Clin. Nutr. 40, 5079–5088 (2021).
Google Scholar
Gonzalez-Palacios, S. et al. Increased ultra-processed food consumption is associated with worsening of cardiometabolic risk factors in adults with metabolic syndrome: longitudinal analysis from a randomized trial. Atherosclerosis 377, 12–23 (2023).
Google Scholar
Pang, T. et al. Ultra-processed food consumption and obesity indicators in individuals with and without type 1 diabetes mellitus: a longitudinal analysis of the prospective Coronary Artery Calcification in Type 1 Diabetes (CACTI) cohort study. Public Health Nutr. 26, 1626–1633 (2023).
Google Scholar
Magalhaes, E. et al. Sex-dependent effects of the intake of NOVA classified ultra-processed foods on syndrome metabolic components in Brazilian adults. Nutrients 14, 3126 (2022).
Google Scholar
Pan, F. et al. Association between ultra-processed food consumption and metabolic syndrome among adults in China – results from the China Jealth and Nutrition Survey. Nutrients 15, 752 (2023).
Google Scholar
Costa, C. S., Del-Ponte, B., Assuncao, M. C. F. & Santos, I. S. Consumption of ultra-processed foods and body fat during childhood and adolescence: a systematic review. Public Health Nutr. 21, 148–159 (2018).
Google Scholar
De Amicis, R. et al. Ultra-processed foods and obesity and adiposity parameters among children and adolescents: a systematic review. Eur. J. Nutr. 61, 2297–2311 (2022).
Google Scholar
Petridi, E. et al. The impact of ultra-processed foods on obesity and cardiometabolic comorbidities in children and adolescents: a systematic review. Nutr. Rev. 82, 913–928 (2024).
Google Scholar
Hall, K. D. et al. Ultra-processed diets cause excess calorie intake and weight gain: an inpatient randomized controlled trial of ad libitum food intake. Cell Metab. 30, 226 (2019).
Google Scholar
Hamano, S. et al. Ultra-processed foods cause weight gain and increased energy intake associated with reduced chewing frequency: a randomized, open-label, crossover study. Diabetes Obes. Metab. 26, 5431–5443 (2024).
Google Scholar
Dicken, S. J. & Batterham, R. L. Ultra-processed food and obesity: what is the evidence? Curr. Nutr. Rep. 13, 23–38 (2024).
Google Scholar
Monteiro, C. A. et al. Ultra-processed foods: what they are and how to identify them. Public Health Nutr. 22, 936–941 (2019).
Google Scholar
Rolls, B. J. The relationship between dietary energy density and energy intake. Physiol. Behav. 97, 609–615 (2009).
Google Scholar
Teo, P. S. et al. Texture-based differences in eating rate influence energy intake for minimally processed and ultra-processed meals. Am. J. Clin. Nutr. 116, 244–254 (2022).
Google Scholar
Forde, C. G., Mars, M. & de Graaf, K. Ultra-processing or oral processing? A role for energy density and eating rate in moderating energy intake from processed foods. Curr. Dev. Nutr. 4, nzaa019 (2020).
Google Scholar
Lasschuijt, M. P., de Graaf, K. & Mars, M. Effects of oro-sensory exposure on satiation and underlying neurophysiological mechanisms – what do we know so far? Nutrients 13, 1391 (2021).
Google Scholar
Teo, P. S., van Dam, R. M., Whitton, C., Tan, L. W. L. & Forde, C. G. Consumption of foods with higher energy intake rates is associated with greater energy intake, adiposity, and cardiovascular risk factors in adults. J. Nutr. 151, 370–378 (2021).
Google Scholar
Uehara, F. et al. Impact of masticatory behaviors measured with wearable device on metabolic syndrome: cross-sectional study. JMIR Mhealth Uhealth 10, e30789 (2022).
Google Scholar
Lasschuijt, M. et al. Speed limits: the effects of industrial food processing and food texture on daily energy intake and eating behaviour in healthy adults. Eur. J. Nutr. 62, 2949–2962 (2023).
Google Scholar
Capuano, E., Oliviero, T., Fogliano, V. & Pellegrini, N. Role of the food matrix and digestion on calculation of the actual energy content of food. Nutr. Rev. 76, 274–289 (2018).
Google Scholar
Zinocker, M. K. & Lindseth, I. A. The Western diet–microbiome–host interaction and its role in metabolic disease. Nutrients 10, 365 (2018).
Google Scholar
Duca, F. A., Waise, T. M. Z., Peppler, W. T. & Lam, T. K. T. The metabolic impact of small intestinal nutrient sensing. Nat. Commun. 12, 903 (2021).
Google Scholar
Wilbrink, J. et al. Review on the regional effects of gastrointestinal luminal stimulation on appetite and energy intake: (pre)clinical observations. Nutrients 13, 1601 (2021).
Google Scholar
Muller, T. D. et al. Glucagon-like peptide 1 (GLP-1). Mol. Metab. 30, 72–130 (2019).
Google Scholar
Wyatt, P. et al. Postprandial glycaemic dips predict appetite and energy intake in healthy individuals. Nat. Metab. 3, 523–529 (2021).
Google Scholar
Oettle, G. J., Emmett, P. M. & Heaton, K. W. Glucose and insulin responses to manufactured and whole-food snacks. Am. J. Clin. Nutr. 45, 86–91 (1987).
Google Scholar
Blaak, E. E. et al. Impact of postprandial glycaemia on health and prevention of disease. Obes. Rev. 13, 923–984 (2012).
Google Scholar
Fardet, A. Minimally processed foods are more satiating and less hyperglycemic than ultra-processed foods: a preliminary study with 98 ready-to-eat foods. Food Funct. 7, 2338–2346 (2016).
Google Scholar
Martinez Steele, E., Raubenheimer, D., Simpson, S. J., Baraldi, L. G. & Monteiro, C. A. Ultra-processed foods, protein leverage and energy intake in the USA. Public Health Nutr. 21, 114–124 (2018).
Google Scholar
Raubenheimer, D. & Simpson, S. J. Protein appetite as an integrator in the obesity system: the protein leverage hypothesis. Philos. Trans. R. Soc. Lond. B Biol. Sci. 378, 20220212 (2023).
Google Scholar
Lynch, C. J. & Adams, S. H. Branched-chain amino acids in metabolic signalling and insulin resistance. Nat. Rev. Endocrinol. 10, 723–736 (2014).
Google Scholar
Handakas, E. et al. Metabolic profiles of ultra-processed food consumption and their role in obesity risk in British children. Clin. Nutr. 41, 2537–2548 (2022).
Google Scholar
Mozaffarian, D. Dietary and policy priorities for cardiovascular disease, diabetes, and obesity: a comprehensive review. Circulation 133, 187–225 (2016).
Google Scholar
Moubarac, J. C. et al. Consumption of ultra-processed foods and likely impact on human health. Evidence from Canada. Public Health Nutr. 16, 2240–2248 (2013).
Google Scholar
Stanhope, K. L. et al. Pathways and mechanisms linking dietary components to cardiometabolic disease: thinking beyond calories. Obes. Rev. 19, 1205–1235 (2018).
Google Scholar
Young, L. R. & Nestle, M. The contribution of expanding portion sizes to the US obesity epidemic. Am. J. Public Health 92, 246–249 (2002).
Google Scholar
Mattes, R. D. Snacking: a cause for concern. Physiol. Behav. 193, 279–283 (2018).
Google Scholar
Gombi-Vaca, M. F., Martinez-Steele, E., Andrade, G. C., Louzada, M. & Levy, R. B. Association between ultra-processed food and snacking behavior in Brazil. Eur. J. Nutr. 63, 1177–1186 (2024).
Google Scholar
Sutton, C. A., Stratton, M., L’Insalata, A. M. & Fazzino, T. L. Ultraprocessed, hyper-palatable, and high energy density foods: prevalence and distinction across 30 years in the United States. Obesity 32, 166–175 (2024).
Google Scholar
Fazzino, T. L., Rohde, K. & Sullivan, D. K. Hyper-palatable foods: development of a quantitative definition and application to the US food system database. Obesity 27, 1761–1768 (2019).
Google Scholar
Fazzino, T. L., Dorling, J. L., Apolzan, J. W. & Martin, C. K. Meal composition during an ad libitum buffet meal and longitudinal predictions of weight and percent body fat change: the role of hyper-palatable, energy dense, and ultra-processed foods. Appetite 167, 105592 (2021).
Google Scholar
Bellitti, J. S., Rohde, K. & Fazzino, T. L. Motives and food craving: associations with frequency of hyper-palatable food intake among college students. Eat. Behav. 51, 101814 (2023).
Google Scholar
Bjorlie, K. et al. Hyper-palatable food consumption during binge-eating episodes: a comparison of intake during binge eating and restricting. Int. J. Eat. Disord. 55, 688–696 (2022).
Google Scholar
Fazzino, T. L., Courville, A. B., Guo, J. & Hall, K. D. Ad libitum meal energy intake is positively influenced by energy density, eating rate and hyper-palatable food across four dietary patterns. Nat. Food 4, 144–147 (2023).
Google Scholar
Small, D. M. & DiFeliceantonio, A. G. Processed foods and food reward. Science 363, 346–347 (2019).
Google Scholar
DiFeliceantonio, A. G. et al. Supra-additive effects of combining fat and carbohydrate on food reward. Cell Metab. 28, 33–44.e3 (2018).
Google Scholar
Gearhardt, A. N. et al. Social, clinical, and policy implications of ultra-processed food addiction. BMJ 383, e075354 (2023).
Google Scholar
Wise, R. A. & Robble, M. A. Dopamine and addiction. Annu. Rev. Psychol. 71, 79–106 (2020).
Google Scholar
Darcey, V. L. et al. Brain dopamine responses to ultra-processed milkshakes are highly variable and not significantly related to adiposity in humans. Cell Metab. 37, 616–628.e5 (2025).
Google Scholar
Kelly, A. L., Baugh, M. E., Oster, M. E. & DiFeliceantonio, A. G. The impact of caloric availability on eating behavior and ultra-processed food reward. Appetite 178, 106274 (2022).
Google Scholar
Schulte, E. M., Avena, N. M. & Gearhardt, A. N. Which foods may be addictive? The roles of processing, fat content, and glycemic load. PLoS ONE 10, e0117959 (2015).
Google Scholar
Rios-Leyvraz, M. & Montez, J. Health effects of the use of non-sugar sweeteners: a systematic review and meta-analysis. WHO www.who.int/publications/i/item/9789240046429 (2022).
Laffitte, A., Neiers, F. & Briand, L. Functional roles of the sweet taste receptor in oral and extraoral tissues. Curr. Opin. Clin. Nutr. Metab. Care 17, 379–385 (2014).
Google Scholar
Chan, C. B., Hashemi, Z. & Subhan, F. B. The impact of low and no-caloric sweeteners on glucose absorption, incretin secretion, and glucose tolerance. Appl. Physiol. Nutr. Metab. 42, 793–801 (2017).
Google Scholar
Whelan, K., Bancil, A. S., Lindsay, J. O. & Chassaing, B. Ultra-processed foods and food additives in gut health and disease. Nat. Rev. Gastroenterol. Hepatol. 21, 406–427 (2024).
Google Scholar
Dalenberg, J. R. et al. Short-term consumption of sucralose with, but not without, carbohydrate impairs neural and metabolic sensitivity to sugar in humans. Cell Metab. 31, 493–502.e7 (2020).
Google Scholar
Bueno-Hernandez, N. et al. Chronic sucralose consumption induces elevation of serum insulin in young healthy adults: a randomized, double blind, controlled trial. Nutr. J. 19, 32 (2020).
Google Scholar
Azad, M. B. et al. Nonnutritive sweetener consumption during pregnancy, adiposity, and adipocyte differentiation in offspring: evidence from humans, mice, and cells. Int. J. Obes. 44, 2137–2148 (2020).
Google Scholar
Martínez Steele, E. & Monteiro, C. A. Association between dietary share of ultra-processed foods and urinary concentrations of phytoestrogens in the US. Nutrients 9, 209 (2017).
Google Scholar
Leitao, A. E. et al. Association between ultra-processed food and flavonoid intakes in a nationally representative sample of the US population. Br. J. Nutr. 131, 1074–1083 (2024).
Google Scholar
Lin, C. Y. et al. Association among acrylamide, blood insulin, and insulin resistance in adults. Diabetes Care 32, 2206–2211 (2009).
Google Scholar
Huang, M., Zhuang, P., Jiao, J., Wang, J. & Zhang, Y. Association of acrylamide hemoglobin biomarkers with obesity, abdominal obesity and overweight in general US population: NHANES 2003-2006. Sci. Total. Env. 631-632, 589–596 (2018).
Google Scholar
Zhang, Y. et al. Exposure to acrylamide and the risk of cardiovascular diseases in the National Health and Nutrition Examination Survey 2003-2006. Env. Int. 117, 154–163 (2018).
Google Scholar
Lee, H. W. & Pyo, S. Acrylamide induces adipocyte differentiation and obesity in mice. Chem. Biol. Interact. 298, 24–34 (2019).
Google Scholar
Thompson, A. K., Minihane, A. M. & Williams, C. M. Trans fatty acids and weight gain. Int. J. Obes. 35, 315–324 (2011).
Google Scholar
Dorfman, S. E. et al. Metabolic implications of dietary trans-fatty acids. Obesity 17, 1200–1207 (2009).
Google Scholar
Srour, B. et al. Ultra-processed foods and human health: from epidemiological evidence to mechanistic insights. Lancet Gastroenterol. Hepatol. 7, 1128–1140 (2022).
Google Scholar
Blackburn, K. & Green, D. The potential effects of microplastics on human health: what is known and what is unknown. Ambio 51, 518–530 (2022).
Google Scholar
Martínez Steele, E., Khandpur, N., da Costa Louzada, M. L. & Monteiro, C. A. Association between dietary contribution of ultra-processed foods and urinary concentrations of phthalates and bisphenol in a nationally representative sample of the US population aged 6 years and older. PLoS ONE 15, e0236738 (2020).
Google Scholar
Dalamaga, M. et al. The role of endocrine disruptors bisphenols and phthalates in obesity: current evidence, perspectives and controversies. Int. J. Mol. Sci. 25, 675 (2024).
Google Scholar
Chassaing, B. & Gewirtz, A. T. Has provoking microbiota aggression driven the obesity epidemic? Bioessays 38, 122–128 (2016).
Google Scholar
Boulange, C. L., Neves, A. L., Chilloux, J., Nicholson, J. K. & Dumas, M. E. Impact of the gut microbiota on inflammation, obesity, and metabolic disease. Genome Med. 8, 42 (2016).
Google Scholar
Cani, P. D. et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 56, 1761–1772 (2007).
Google Scholar
Fernandes, A. E. et al. Differences in the gut microbiota of women according to ultra-processed food consumption. Nutr. Metab. Cardiovasc. Dis. 33, 84–89 (2023).
Google Scholar
Dapa, T., Ramiro, R. S., Pedro, M. F., Gordo, I. & Xavier, K. B. Diet leaves a genetic signature in a keystone member of the gut microbiota. Cell Host Microbe 30, 183–199.e10 (2022).
Google Scholar
Lopez-Moreno, J. et al. Effect of dietary lipids on endotoxemia influences postprandial inflammatory response. J. Agric. Food Chem. 65, 7756–7763 (2017).
Google Scholar
Naimi, S., Viennois, E., Gewirtz, A. T. & Chassaing, B. Direct impact of commonly used dietary emulsifiers on human gut microbiota. Microbiome 9, 66 (2021).
Google Scholar
De Siena, M. et al. Food emulsifiers and metabolic syndrome: the role of the gut microbiota. Foods 11, 2205 (2022).
Google Scholar
Laster, J., Bonnes, S. L. & Rocha, J. Increased use of emulsifiers in processed foods and the links to obesity. Curr. Gastroenterol. Rep. 21, 61 (2019).
Google Scholar
Chassaing, B. et al. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature 519, 92–96 (2015).
Google Scholar
Chassaing, B. et al. Randomized controlled-feeding study of dietary emulsifier carboxymethylcellulose reveals detrimental impacts on the gut microbiota and metabolome. Gastroenterology 162, 743–756 (2022).
Google Scholar
Im, E., Riegler, F. M., Pothoulakis, C. & Rhee, S. H. Elevated lipopolysaccharide in the colon evokes intestinal inflammation, aggravated in immune modulator-impaired mice. Am. J. Physiol. Gastrointest. Liver Physiol. 303, G490–G497 (2012).
Google Scholar
Nettleton, J. E., Reimer, R. A. & Shearer, J. Reshaping the gut microbiota: impact of low calorie sweeteners and the link to insulin resistance? Physiol. Behav. 164, 488–493 (2016).
Google Scholar
Suez, J. et al. Personalized microbiome-driven effects of non-nutritive sweeteners on human glucose tolerance. Cell 185, 3307–3328.e19 (2022).
Google Scholar
Serrano, J. et al. High-dose saccharin supplementation does not induce gut microbiota changes or glucose intolerance in healthy humans and mice. Microbiome 9, 11 (2021).
Google Scholar
Arnold, A. R. & Chassaing, B. Maltodextrin, modern stressor of the intestinal environment. Cell Mol. Gastroenterol. Hepatol. 7, 475–476 (2019).
Google Scholar
Makki, K., Deehan, E. C., Walter, J. & Backhed, F. The impact of dietary fiber on gut microbiota in host health and disease. Cell Host Microbe 23, 705–715 (2018).
Google Scholar
Sonnenburg, E. D. & Sonnenburg, J. L. Starving our microbial self: the deleterious consequences of a diet deficient in microbiota-accessible carbohydrates. Cell Metab. 20, 779–786 (2014).
Google Scholar
Mialon, M., Corvalan, C., Cediel, G., Scagliusi, F. B. & Reyes, M. Food industry political practices in Chile: “the economy has always been the main concern”. Glob. Health 16, 107 (2020).
Mialon, M. & Gomes, F. D. S. Public health and the ultra-processed food and drink products industry: corporate political activity of major transnationals in Latin America and the Caribbean. Public Health Nutr. 22, 1898–1908 (2019).
Google Scholar
Popkin, B. M. et al. Towards unified and impactful policies to reduce ultra-processed food consumption and promote healthier eating. Lancet Diabetes Endocrinol. 9, 462–470 (2021).
Google Scholar
Gupta, S., Hawk, T., Aggarwal, A. & Drewnowski, A. Characterizing ultra-processed foods by energy density, nutrient density, and cost. Front. Nutr. 6, 70 (2019).
Google Scholar
Vandevijvere, S., Pedroni, C., De Ridder, K. & Castetbon, K. The cost of diets according to their caloric share of ultraprocessed and minimally processed foods in Belgium. Nutrients 12, 2787 (2020).
Google Scholar
Moubarac, J. C. et al. International differences in cost and consumption of ready-to-consume food and drink products: United Kingdom and Brazil, 2008-2009. Glob. Public Health 8, 845–856 (2013).
Google Scholar
Meyer, I. Interactive map: tracking state food chemical regulation in the U.S. EWG www.ewg.org/news-insights/news/2025/03/interactive-map-tracking-state-food-chemical-regulation-us (2025).
Hagerman, C. J., Hong, A. E., Jennings, E. & Butryn, M. L. A pilot study of a novel dietary intervention targeting ultra-processed food intake. Obes. Sci. Pract. 10, e70029 (2024).
Google Scholar
Anastasiou, K. et al. From harmful nutrients to ultra-processed foods: exploring shifts in ‘foods to limit’ terminology used in national food-based dietary guidelines. Public Health Nutr. 26, 2539–2550 (2023).
Google Scholar
Arenas, A. B. et al. Guías alimentarias y de actividad física en contexto de sobrepeso y obesidad en la población Mexicana. INSP www.insp.mx/resources/images/stories/2015/Noticias/Nutricion_y_Salud/Docs/151118_guias_alimentarias.pdf (2015).
Health Protection Agency. Food based dietary guidelines for Maldives. Health Protection Agency health.gov.mv/storage/uploads/50wNJEwA/ydrp2hei.pdf (2017).
National Coordinating Committee on Food and Nutrition. Malaysian dietary guidelines 2020. Ministry of Health Malaysia hq.moh.gov.my/nutrition/wp-content/uploads/2024/03/latest-01.Buku-MDG-2020_12Mac2024.pdf (2020).
Crosbie, E. et al. A policy study on front-of-pack nutrition labeling in the Americas: emerging developments and outcomes. Lancet Reg. Health Am. 18, 100400 (2023).
Google Scholar
Popkin, B. M. & Ng, S. W. The nutrition transition to a stage of high obesity and noncommunicable disease prevalence dominated by ultra-processed foods is not inevitable. Obes. Rev. 23, e13366 (2022).
Google Scholar
Taylor, L. Colombia introduces Latin America’s first junk food tax. BMJ 383, 2698 (2023).
Google Scholar
National Education Development Fund. Resolution no. 6, of May 8, 2020. gov.br www.gov.br/fnde/pt-br/acesso-a-informacao/legislacao/resolucoes/2020/resolucao-no-6-de-08-de-maio-de-2020/view (2020).
Ministério dos Direitos Humanos e da Cidadania. Governo federal reduz para 15% o limite de alimentos processados e ultraprocessados nas escolas públicas. Gov.br (2025).
link