Preclinical atherosclerosis and prediabetes: a cross-sectional metabolic assessment in apparently healthy population | Cardiovascular Diabetology

The link between preclinical atherosclerosis and prediabetes in study sample
Among individuals’ prediabetes-free, 19.5% were found to have preclinical atherosclerosis, compared to 30.8% among those with prediabetes. The difference between the groups was statistically significant (p = 0.006). The fundamental clinical characteristics of the study cohort (n = 447, 44.7% males) are presented in Table 1. The selected population sample had a mean age of 39.7 ± 9.6 years. The median BMI was 24.77 kg/m2, indicating that most individuals were within the normal weight range. On average, both the lipid profile and glucose metabolism parameters were within normal ranges. Additionally, Supplementary Table 1 presents the characteristics of the selected cohort stratified by the presence or absence of prediabetes and preclinical atherosclerosis. Group 1 includes individuals with neither condition (n = 48), group 2 comprises participants with prediabetes only (n = 62), group 3 includes those with preclinical atherosclerosis only (n = 198) and group 4 consists of individuals presenting with both prediabetes and subclinical atherosclerosis (n = 139). This classification enabled the assessment of metabolic differences associated with each condition independently, as well as their potential synergistic effects.
The assessment of effects of preclinical atherosclerosis and prediabetes on metabolites
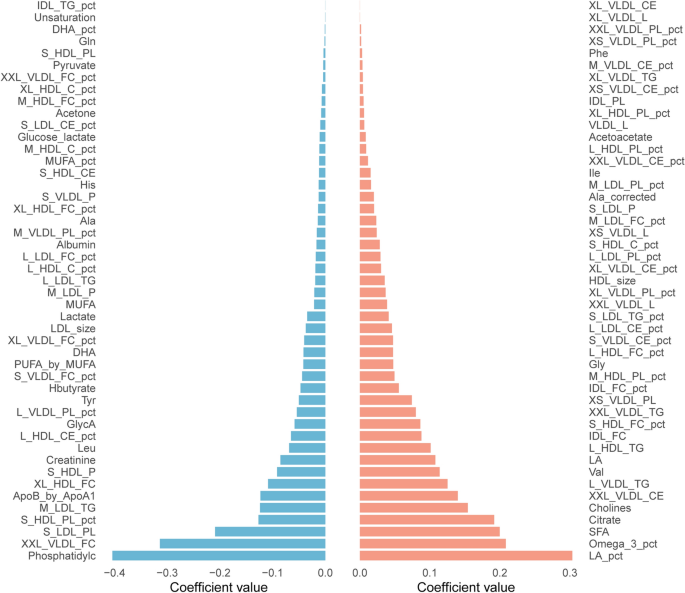
Figure 1 and Supplementary Table 2 present the results of a two-way ANOVA evaluating the associations between individual metabolite concentrations and two clinical conditions: atherosclerosis and prediabetes, including their interaction effect. After Benjamini–Hochberg false discovery rate (FDR) adjustment, a total of 20 metabolites were found to be significantly associated exclusively with prediabetes, 4 exclusively with preclinical atherosclerosis, and another 4 were significantly associated with both conditions, suggesting overlapping metabolic alterations. Only 1 metabolite, TMAO, showed a significant association with the interaction term. Among the metabolites with the strongest associations, Glu, CE(20:5) (cholesteryl ester of eicosapentaenoic acid), CE(20:3), and CE(18:3) were significantly associated with both atherosclerosis and prediabetes. Dehydroepiandrosterone sulfate (DHEAS), indole-3-propionic acid (IPA), and CE(16:1) were primarily associated with preclinical atherosclerosis. Lac, L-Ala, docosahexaenoic acid (DHA), eicosapentaenoic acid (EPA), and dihomo-gamma-linolenic acid (DGLA) were among the most strongly associated with prediabetes.
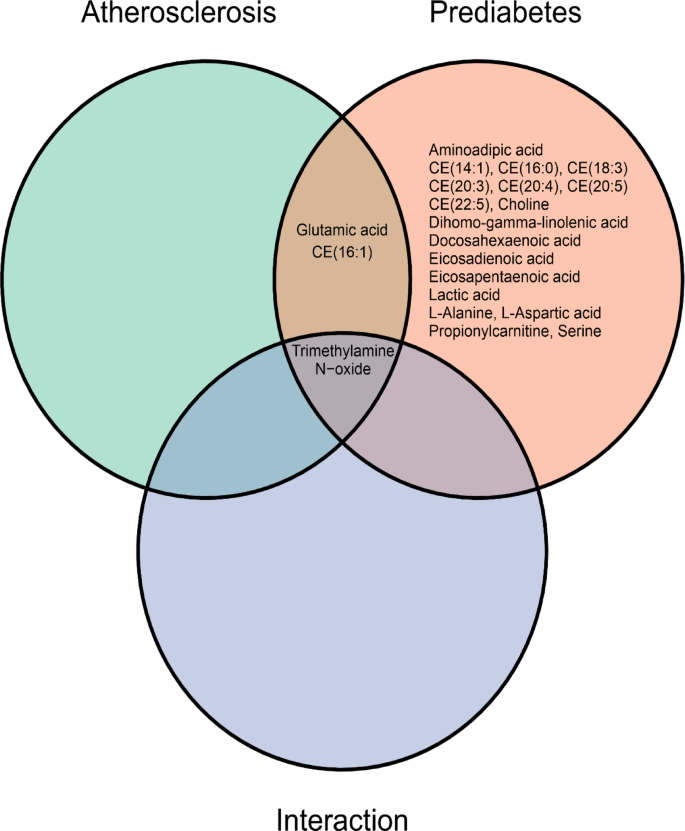
Venn diagram illustrating the number of metabolites significantly associated with atherosclerosis status, prediabetes status as identified by two-way ANOVA. Trimethylamine N-oxide (TMAO) was not directly correlated with prediabetes and preclinical atherosclerosis, but was associated with their interaction.
A summary of the results of linear regression models assessing the independent and interaction effects of preclinical atherosclerosis, prediabetes on individual metabolite concentrations, adjusted for age and sex (including only those with statistically significant associations), is presented in Table 2. After applying false discovery rate (FDR) correction for multiple comparisons, no metabolites demonstrated statistically significant associations with preclinical atherosclerosis alone. In contrast, seven metabolites showed significant associations with prediabetes, including L-Ala, Glu, and Lac, which exhibited the strongest associations with dysglycemia. Additional metabolites, such as DHEAS, DGLA, and EPA, also remained significant following adjustment for sex and age. Supplementary Table 3 presents additional linear regression models adjusted not only for age and sex but also for established cardiovascular risk factors, including BMI, current smoking status, fasting glucose concentration, HOMA-IR, as well as LDL-C and HDL-C levels. Notably, TMAO remained a statistically significant predictor even after adjustment for these covariates, highlighting its potential role as a key metabolic mediator in the interplay between preclinical atherosclerosis and prediabetes.
Notably, TMAO was the only metabolite demonstrating statistically significant association with both prediabetes and the interaction between atherosclerosis and prediabetes, suggesting a combined, non-additive effect. This interaction was visualized on a violin plot (Fig. 2), illustrating the differing effects of prediabetes across atherosclerosis strata. Post hoc stratified analysis (adjusted for age and sex) revealed that in individuals without atherosclerosis, TMAO levels were significantly higher in those with prediabetes compared to prediabetes-free participants (β = 1.11, p = 0.0235). In contrast, among individuals with atherosclerosis, TMAO levels did not differ significantly by prediabetes status (β =–2.69, p = 0.18). These findings imply a potential context-dependent effect of prediabetes on TMAO, modulated by the presence of preclinical atherosclerosis.
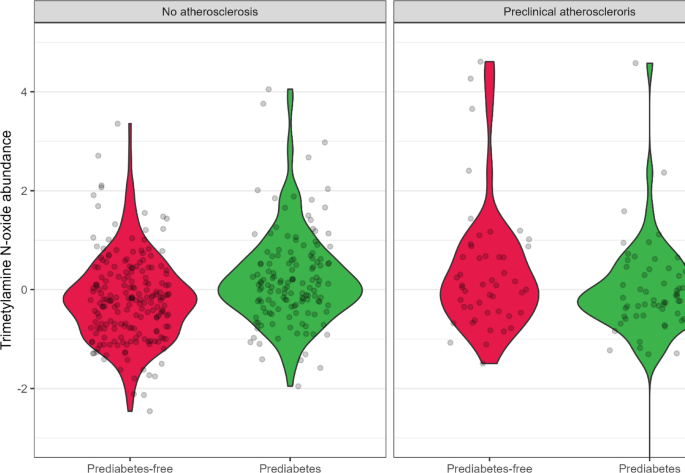
Trimethylamine N-oxide levels in relation to prediabetes and preclinical atherosclerosis
The evaluation of the prediabetes and preclinical atherosclerosis effect on the modulation of metabolite profiles
The results of the two-way ANOVA assessing the independent and interaction effects of prediabetes and preclinical atherosclerosis on metabolite sums and ratios are presented in Fig. 3 and Supplementary Table 4. Significant main effects of atherosclerosis were predominantly observed in lipid-related parameters, including the ratio of lysophosphatidylcholines (LPCs) containing polyunsaturated fatty acids (PUFAs) to those containing saturated fatty acids (SFAs), the sum of steroid hormones, and selected classes of cholesteryl esters—specifically those containing monounsaturated (MUFA-CEs) and long-chain fatty acids (LCFA-CEs).
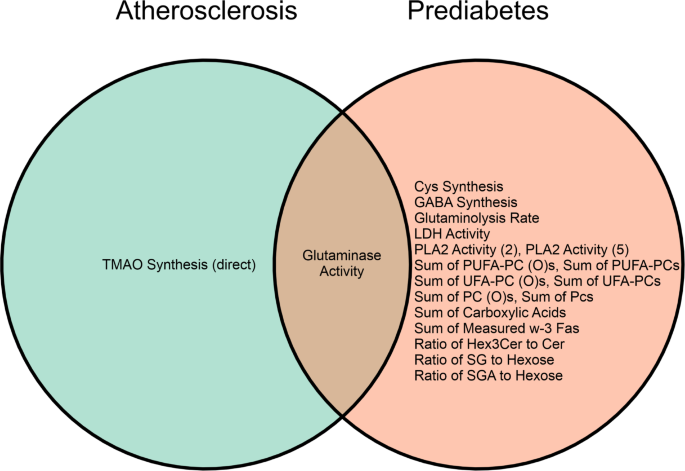
Venn diagram of associations between metabolite sums and ratios with preclinical atherosclerosis, prediabetes, based on ANOVA results
In contrast, the main effects of prediabetes showed a broader metabolic footprint, with significant associations related to AA metabolism and lipid signalling. These included elevated sums of aromatic and sulfur-containing amino acids (AAs), along with increased activities of phospholipase A2 (PLA2) and indoleamine 2,3-dioxygenase (IDO). Additionally, higher levels of very-long-chain fatty acid dihydroceramides (VLCFA-DH-Cer) and the overall pool of dihydroceramides (DH-Cer) were observed. Perturbations in the profiles of unsaturated phosphatidylcholines (UFA-PCs) and ether-linked phosphatidylcholine species (PC (O)s), including both unsaturated (UFA-PC (O)) and saturated (SFA-PC (O)) variants, were also detected. Furthermore, the ratio of saturated galactosylceramides (SG) to hexoses and concentrations of measured omega-3 fatty acids (ω-3 FAs) were significantly elevated in individuals with prediabetes. The sum of carboxylic acids and the calculated glutaminolysis rate were also significantly associated with dysglycemia-related metabolic disruptions.
Several metabolic features were found to be dysregulated in both prediabetes and preclinical atherosclerosis. Glutaminase activity, assessed as Glu/Gln ratio [17] and glutathione (GSH) pathway were significantly associated with both conditions. Furthermore, multiple PLA2 activities, specifically PLA2 Activity (3) and PLA2 Activity (5), indicate shared activation of phospholipase A2-mediated pathways. Other shared features included the sum of PUFA-containing phosphatidylcholines (PUFA-PCs), ether-linked PUFA-PC (O)s, and very-long-chain fatty acid cholesteryl esters (VLCFA-CEs).
Table 3 provides a summary of the linear regression analyses, which evaluated associations between selected metabolic features (sums and ratios) and three factors: prediabetes, preclinical atherosclerosis, and their interaction, with adjustments for age and sex. Glutaminase activity emerged as the only shared predictor of both prediabetes and atherosclerosis, highlighting a potentially convergent glutamine-related metabolic mechanism. In contrast, the glutaminolysis rate, the sum of carboxylic acids, and the ratio of SG to hexose were uniquely associated with prediabetes. No distinct metabolic predictors were identified exclusively for atherosclerosis or for the interaction term. Supplementary Table 5 provides a summary of linear regression models in which CVR factors were reintroduced as covariates. Following adjustment for BMI and current smoking status, glutaminase activity continued to demonstrate a statistically significant association, indicating its potential independent contribution to the observed metabolic alterations.
The correlations between metabolites and clinical parameters
Figure 4 presents the Spearman correlation matrix between selected metabolite levels and clinical parameters, including lipid profile components, glucose metabolism indicators, and preclinical atherosclerosis parameters. Only statistically significant correlations (FDR-adjusted p < 0.05 and|ρ|≥ 0.1) are shown. Among the most prominent observations, cholesteryl ester (CE) (20:4) exhibited moderate positive correlations with multiple glycaemic and insulinemic parameters, including fasting glucose, fasting insulin, and baseline C-peptide levels. Fatty acids also showed a positive correlation with parameters of glucose metabolism, especially postprandial. Glutamic acid (Glu) was consistently positively associated with both glucose and insulin concentrations across all assessed time points, suggesting a potential role in glucose homeostasis. In contrast, serine demonstrated inverse correlations with several metabolic markers, most notably with 120-min post-load glucose and C-peptide levels, as well as TG. Among sphingolipids, SM C16:1 showed positive associations with TC and its fractions and IMT, suggesting a possible role in early atherosclerotic processes. HDL-C fraction, in contrast to other lipemic parameters, negatively correlated with a broad spectrum of metabolites, including glutamic acid, aminoadipic acid, lactic acid (Lac), L-alanine (L-Ala), and L-cysteine (L-Cys).
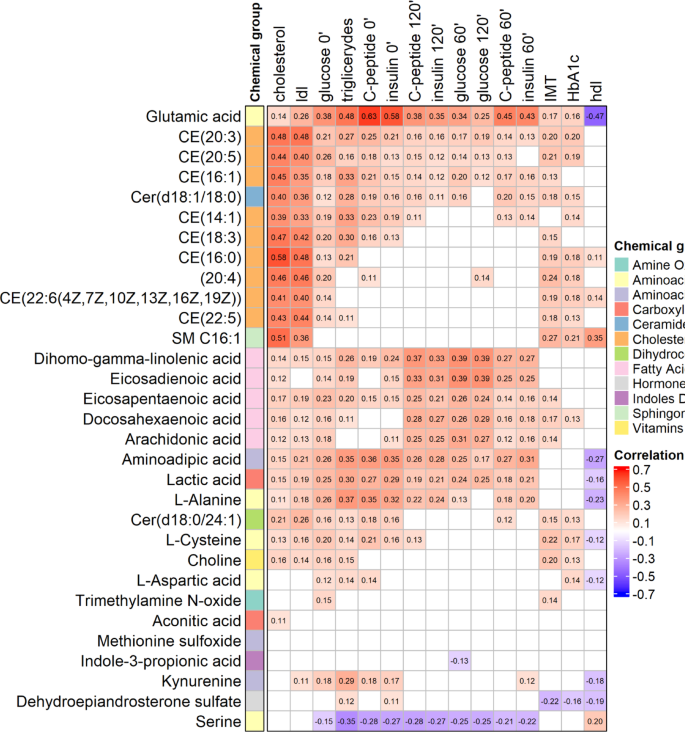
Spearman correlation heatmap between serum metabolites and cardiometabolic biomarkers. Each cell represents the Spearman correlation coefficient between a metabolite and a clinical variable. The color scale ranges from red (positive correlation) to blue (negative correlation), with white indicating values near zero or associations that did not reach statistical significance. Only statistically significant correlations (FDR-adjusted p < 0.05) with absolute correlation coefficients ≥ 0.1 are shown. Exact Spearman rho values are indicated in each cell
Metabolite set enrichment analysis
Figure 5 and Supplementary Table 6 present the results of a Kyoto Encyclopedia of Genes and Genomes (KEGG) [18] metabolic pathway enrichment analysis (MSEA) conducted to identify metabolic pathways significantly associated with preclinical atherosclerosis. The analysis was performed based on differentially abundant metabolites and includes multiple testing correction using the false discovery rate (FDR) approach. Pathways are ranked according to their enrichment ratio (logarithmic scale) and color-coded by adjusted p-values. The most significantly enriched pathways (FDR < 0.001) include glutathione metabolism, steroid biosynthesis, thiamine metabolism, butanoate metabolism, and nitrogen metabolism. This KEGG-based pathway enrichment analysis highlights a distinct metabolic signature associated with preclinical atherosclerosis, implicating redox balance (glutathione), lipid synthesis (steroid biosynthesis), and energy-related pathways (thiamine and butanoate metabolism). These findings suggest that early stages of atherosclerosis are linked to oxidative stress responses, disturbances in lipid biosynthesis, and alterations in energy metabolism. Additional significantly enriched pathways include taurine and hypotaurine metabolism, histidine metabolism, arginine and proline metabolism, alanine, aspartate, and glutamate metabolism, and cysteine and methionine metabolism, indicating broader disruptions in amino acid and nitrogen metabolism. Collectively, these results highlight the involvement of redox regulation, amino acid utilization, and bioenergetic pathways in the metabolic signature of preclinical atherosclerosis.
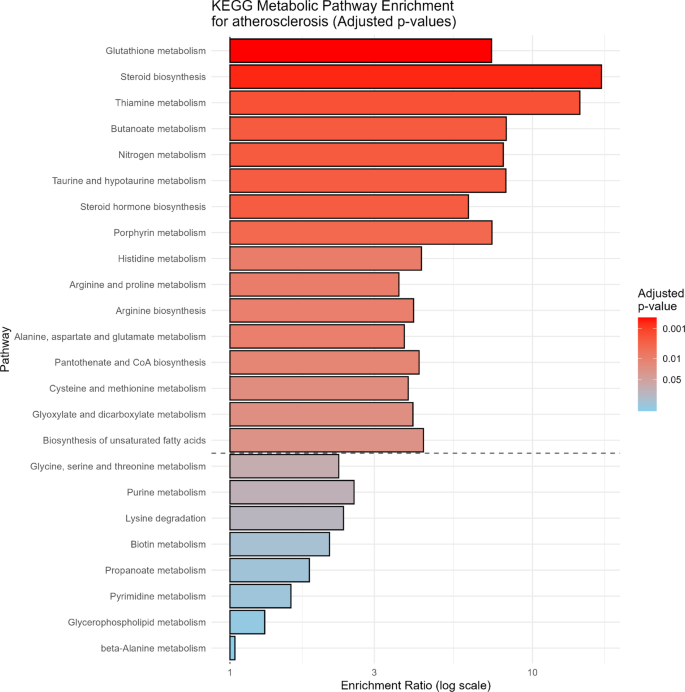
KEGG Pathway Enrichment Analysis for preclinical atherosclerosis
Simultaneously, Fig. 6 and Supplementary Table 7 display results from a KEGG MSEA investigating metabolic alterations associated with prediabetes, based on significantly altered metabolites. A number of pathways show highly significant enrichment (adjusted p < 0.001), including biosynthesis of unsaturated fatty acids, alanine, aspartate, and glutamate metabolism, histidine metabolism, butanoate metabolism, as well as nitrogen metabolism. Additional enriched pathways include glutathione and porphyrin metabolism, both of which are involved in oxidative stress regulation. These results highlight a broad metabolic disruption in prediabetes, involving not only energy metabolism but also amino acid turnover and nitrogen balance.
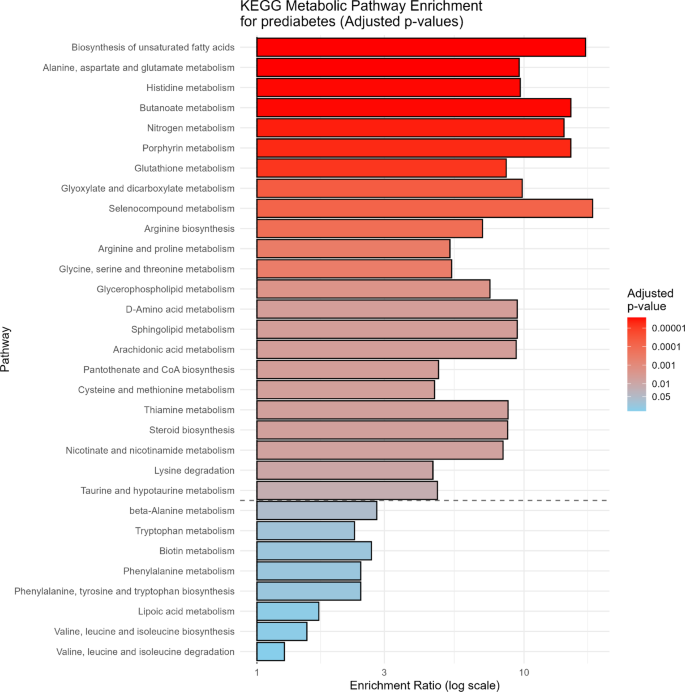
KEGG Pathway Enrichment Analysis for prediabetes
MSEA using the Small Molecule Pathway Database (SMPDB) [19] also revealed distinct yet overlapping profiles in individuals with atherosclerosis and prediabetes. Figure 7 and Supplementary Table 8 showed that in preclinical atherosclerosis, the most significantly enriched pathways included cysteine metabolism, glutathione metabolism, androgen and estrogen metabolism, folate metabolism, and pathways related to AA metabolism such as pantothenate and CoA biosynthesis, glutamate metabolism, and amino sugar metabolism. Lipid-related pathways, such as arachidonic acid and alpha-linolenic acid metabolism, were also significantly represented. In contrast, prediabetes was marked by robust enrichment of the glucose–alanine cycle, glutathione metabolism, alanine metabolism, arachidonic acid metabolism, and glutamate metabolism, along with key alterations in the Warburg effect, tyrosine metabolism, and folate metabolism (Fig. 8 and Supplementary Table 9). Both conditions demonstrated significant enrichment of several shared pathways, including glutathione metabolism, glutamate metabolism, folate metabolism, the Warburg effect, glucose-alanine metabolism, malate-aspartate shuttle, and arachidonic acid metabolism, indicating common metabolic perturbations associated with oxidative stress, inflammation, and mitochondrial dysfunction. In particular, the involvement of tryptophan and glycerophospholipid metabolism highlights the importance of immune-metabolic crosstalk and membrane lipid remodeling in the pathogenesis of these interconnected conditions.
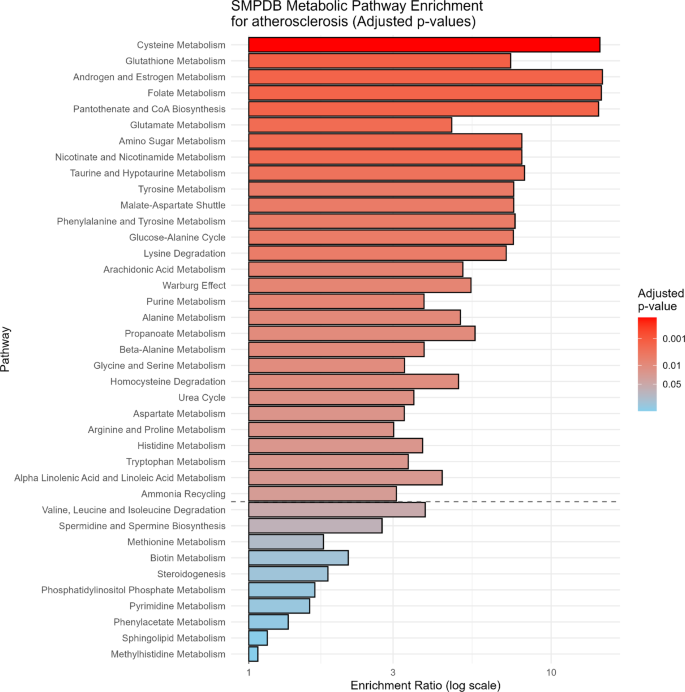
SMPDB Pathway Enrichment Analysis for preclinical atherosclerosis
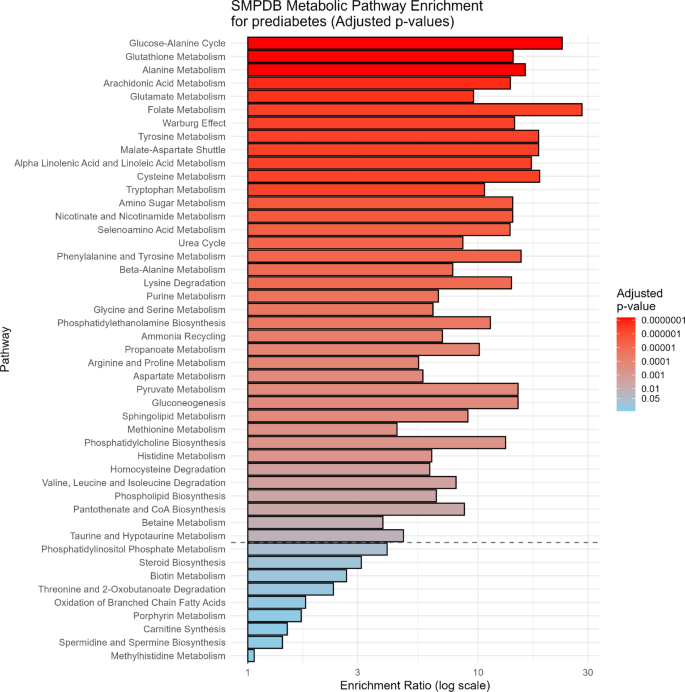
SMPDB Pathway Enrichment Analysis for prediabetes
link