Metabolic impact of dietary glycine supplementation in individuals with severe obesity
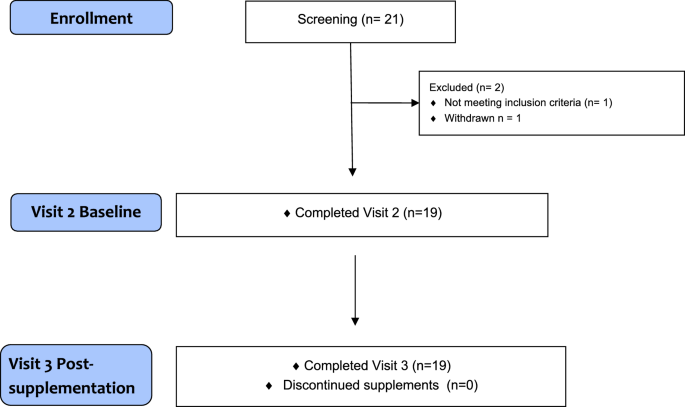
The primary aim of this study was to determine whether oral glycine supplementation would correct its deficiency in individuals with severe obesity and to evaluate its impact on the metabolic pathways that are dependent on glycine availability, including the glycine conjugation detoxification pathway, GSH synthesis, and the 1-carbon cycle. We found that glycine supplementation significantly increased the plasma concentration of glycine and enhanced the formation and urinary excretion of several acylglycines. Glycine supplementation lowered plasma triglyceride, hepatic aminotransferases, and the GSG index, a biomarker of MASLD, without changes in body weight, insulin resistance, energy expenditure, or whole-body substrate oxidation rates. Plasma concentration of several metabolites involved in the 1-carbon cycle increased post-treatment, but the treatment did not affect RBC GSH or oxidative stress marker concentrations. These findings indicate that oral glycine supplementation corrected glycine deficiency in individuals with severe obesity, which in turn increased flux through the glycine conjugation pathway and the one-carbon cycle. Most importantly, these changes were associated with marked improvements in several biomarkers of liver health.
Glycine is conventionally regarded as a nutritionally non-essential amino acid. However, glycine deficiency develops when the human body cannot synthesize adequate glycine to meet its metabolic demand. In the late stages of pregnancy, slower glycine de novo synthesis limits the ability of adolescent mothers to maintain glycine flux and compromises fetal growth22. Similarly, we found that a diminished de novo glycine synthesis rate led to glycine deficiency in individuals with severe obesity3. These findings suggest simple measures, such as dietary supplementation, could be used to correct glycine deficiency. Glycine is the smallest amino acid and is rapidly absorbed. A single dose of dietary glycine raises plasma glycine concentration after 10 min23, and earlier clinical trials showed that two weeks of dietary glycine supplementation at a similar dose to what was used in this study significantly raised glycine levels in individuals with glycine deficiency from HIV infection18, uncontrolled diabetes17, and old age19,24. However, glycine supplementation has not been attempted in individuals with severe obesity, and the amount or duration of glycine required to correct glycine deficiency in individuals with severe obesity is unknown. Participants in this study consumed an average of 83 g of protein, translating to approximately 2.5 g of glycine per day. We demonstrated that treatment with glycine supplements at 100 mg/kg/day (4 times the amount of the participants’ dietary glycine intake) for two weeks raised plasma glycine concentration by 35%. This finding confirms glycine de novo synthesis is impaired in the obese state and should be considered a conditionally essential amino acid. In addition, because the study subjects were glycine deficient, the additional 1.9 g of nitrogen intake from glycine supplementation was unlikely to have resulted in a state of positive nitrogen balance and increased urine nitrogen excretion.
Glycine participates in multiple metabolic reactions and is involved in the human body’s detoxification system. AcylCoAs, produced during cellular substrate metabolism, become metabolically toxic at high levels and must be cleared to maintain normal cellular function. The glycine conjugation pathway is a phase II detoxification reaction catalyzed by the mitochondrial enzyme glycine-N-acyltransferase. This reaction involves transferring the acyl group from acyl-CoA esters to the N-terminus of glycine to form N-acylglycines, which are cleared from the human body via urinary excretion. In obesity, accelerated substrate turnover rates result in a huge influx of substrates that require oxidation and clearance in the mitochondria. However, when the influx of substrates exceeds mitochondrial oxidative capacity, these intermediates accumulate in the liver as acylCoAs. This pathway serves as an alternative elimination route, but the short supply of glycine in the obese state may impair glycine conjugation and compromise this detoxification system. Several human and rodent studies found that obesity-associated glycine deficiency was associated with decreased urine excretion of acylglycines derived from acetylCoA and BCAA oxidation intermediates21,24,25. Using stable-isotope tracers, we recently confirmed that obesity-associated glycine deficiency compromised the synthesis and clearance of endogenous and xenobiotic acylCoAs as acylglycines3.
The metabolic consequence of impaired detoxification via this glycine-dependent pathway is unclear. However, since this reaction occurs primarily in the liver, glycine deficiency could impede the elimination of excess acylCoAs from the liver of individuals with obesity and contribute to MASLD pathogenesis. This also means that interventions that enhance this elimination pathway could be used to treat MASLD. In this study, we demonstrated that the correction of glycine deficiency with dietary supplements increased the urinary excretion of acylglycine derived from the intermediates of the BCAAs (isobutyrylglycine, tigylglycine, and isovalerylglycine) and fatty acid oxidation (hexanoylglycine). The higher urinary excretion of isobutyrylglycine, tigylglycine, and isovalerylglycine also suggests that glycine supplementation increases hepatic BCAA catabolism. However, the plasma BCAA concentrations were not affected. In addition to the liver, the plasma concentration of BCAAs is also determined by the release and uptake of BCAAs from other organs, such as skeletal muscle and adipose tissue26. Thus, the lack of post-treatment alteration in plasma BCAA concentration could be attributed to the effect of glycine supplementation on BCAA metabolism in different organs. These tissue-specific effects were observed in mice receiving glycine supplementation27, but it needs to be further investigated in humans.
Our findings were consistent with an earlier study that showed greater excretion of acylglycines and reductions in hepatic short- and intermediate-chain acylCoAs without altering blood BCAA levels in Zucker Fatty Rats receiving dietary glycine supplements27. We also detected statistically significant post-treatment reductions in plasma triglyceride, hepatic transaminases, and the GSG index. We did not perform any imaging or histological examination of liver tissue. However, these parameters are markers of MASLD28,29, and the GSG index can predict the presence of biopsy-proven hepatic fibrosis30. Thus, we hypothesize that glycine supplementation reduces hepatic steatosis and inflammation by enhancing the elimination of excess acylCoAs in the liver as acylglycines.
Glycine supplements’ ability to reduce MASLD severity in individuals with obesity needs additional validation. Nonetheless, the therapeutic potential of glycine as a novel treatment for MASLD is supported by recent preclinical studies that demonstrated the ability of glycine-based supplements to reduce hepatic fat, inflammation, and fibrosis in rodents and non-human primates with MASLD31,32. However, these improvements were attributed to glycine’s ability to enhance GSH synthesis, redox balance, and fatty acid oxidation. GSH is the most abundant intracellular antioxidant synthesized using glycine, cysteine, and glutamine, and a lack of GSH could contribute to oxidative stress and the development of obesity-associated complications. Glycine may be rate-limiting for GSH synthesis9, and we recently reported impaired RBC GSH synthesis in individuals with obesity-associated glycine deficiency8. Thus, glycine supplementation could increase GSH availability and improve redox balance. In individuals with elevated oxidative stress due to aging, HIV infection, and uncontrolled diabetes, glycine and cysteine supplementation raised GSH concentration and lowered oxidative stress17,18,20. By contrast, in our study, RBC GSH and plasma MDA concentrations did not improve following glycine supplementation. Glycine utilization for GSH synthesis may depend on individuals’ redox status. In the largest randomized controlled trial using glycine (and cystine) supplementation, treatment of 114 healthy older adults also did not improve RBC GSH or reduce oxidative stress. However, GSH levels did increase in a subset of participants with low GSH and high MDA levels at baseline19. Our study participants were relatively young and free from any serious chronic diseases. Therefore, oxidative stress levels and GSH demand in these participants may not be enough to divert glycine supply for GSH synthesis.
With glycine supplementation, we observed a significant increase in plasma serine by 17%. Glycine and serine are interconvertible via the serine hydroxymethyltransferase (SHMT), and the increase in serine reflects the rapid conversion of glycine to serine33,34. In addition, total cysteine, folate, and homocysteine concentrations were significantly higher post-treatment. Glycine and serine are principal donors of single-carbon units to the one-carbon metabolism cycle, a key metabolic process in the human body that involves folate-mediated movement of single-carbon units for methylation, redox, and biosynthetic reactions15. Homocysteine, as a component of the one-carbon cycle, can be converted to methionine via the trans-methylation pathway or cysteine via the trans-sulfuration pathway15,35. Thus, we interpret the higher post-treatment values of serine, folate, homocysteine, and cysteine as the consequence of the greater entry of one-carbon units, hence, flux along the one-carbon cycle. As the liver is also the major site for one-carbon metabolism, which is dysregulated in MASLD16, glycine treatment may benefit liver health through this mechanism. This notion is supported by a recent pre-clinical study that showed that the downregulation of hepatic SHMT2 in mice led to changes in hepatic glycine and serine content, which reduced hepatic methylation potential and modulated hepatic fat synthesis, fibrosis, and inflammation36.
The slight elevation of homocysteine observed in our study is unexpected since serine and folate lower homocysteine levels in individuals with hyperhomocysteinemia37,38. Higher plasma homocysteine levels have been associated with a greater incidence of coronary heart disease39. The increase in plasma homocysteine concentration in this study was marginal at 0.6 µmol/L, and the causal relationship between hyperhomocysteinemia and adverse health outcomes is still uncertain40,41. Nonetheless, to truly understand the safety of consuming dietary glycine supplements at the current dose, future long-term studies will be needed to confirm the effects of dietary glycine supplements on homocysteine levels and adverse clinical outcomes. Further, dedicated experiments are also required to examine its metabolic impact on various methylation reactions.
We did not detect significant body weight or composition changes in the study participants following glycine supplementation. This is unsurprising as they maintained the same dietary energy and macronutrient intake throughout the study. Furthermore, indirect calorimetry measurements and plasma acylcarnitine profiling indicate that glycine supplementation did not shift energy expenditure or metabolic fuel selection. Glycine correlates inversely with insulin resistance and diabetes risk. However, the ability of glycine to modulate glucose regulation remains unresolved. While some human studies have reported the potential effects of a single dose of oral glycine to reduce glucose excursion or increase insulin secretion23,42, other studies using oral or intravenous glycine failed to demonstrate any response43,44. Similarly, individuals with metabolic syndrome treated with high doses of oral glycine for 3 months did not demonstrate any significant change in plasma insulin concentration45. Our study was consistent with these findings as we did not detect any statistically significant post-treatment changes in glucose and insulin values at fasting or following glucose challenge.
This study demonstrates that glycine supplementation in individuals with severe obesity enhances the glycine conjugation detoxification pathway and one carbon cycle. These two metabolic reactions occur primarily in the liver, and the associated improvement in MASLD markers suggests glycine supplements could be developed as a low-risk, cost-effective treatment for NAFD. However, there are several limitations. We showed that dietary glycine supplementation increased plasma glycine concentration by 35%, achieving a value similar to healthy normal-weight individuals (~ 200 µmol/L) from another study3. However, our study did not include a healthy control group to confirm the correction of glycine deficiency. Further, as an exploratory study with a limited sample size and short study duration, this study is designed to demonstrate the ability of dietary glycine to raise post-treatment plasma glycine concentration and not improvements in clinical parameters or outcomes. Our study also did not perform liver imaging or histological examination of liver tissue, but MASLD is almost always present in patients with severe obesity46. Previous glycine supplementation trials were also limited by their study design, sample size, and duration, and have yet demonstrated a definite improvement in clinical outcomes47,48. Therefore, future randomized controlled trials with an adequately powered sample size and longer study duration will be necessary to confirm the clinical efficacy and safety of dietary glycine supplementation. Specifically, liver imaging or biopsy will be required to verify the effectiveness of dietary glycine supplementation in treating MASLD. Additional studies will also be required to mechanistically link enhanced urine acylglycine excretion and one carbon cycle activity with MASLD pathogenesis. Further, the downstream metabolic sequelae from enhanced acylglycine excretion and 1-carbon cycle activity in other organs and tissues should be explored in future efforts.
In conclusion, we found that dietary glycine supplements corrected obesity-associated glycine deficiency, increased urine acylglycine excretion, and plasma metabolites associated with one-carbon metabolism. The post-treatment improvement in hepatic markers suggests that glycine supplements should be investigated as a potential novel treatment for MASLD.
link