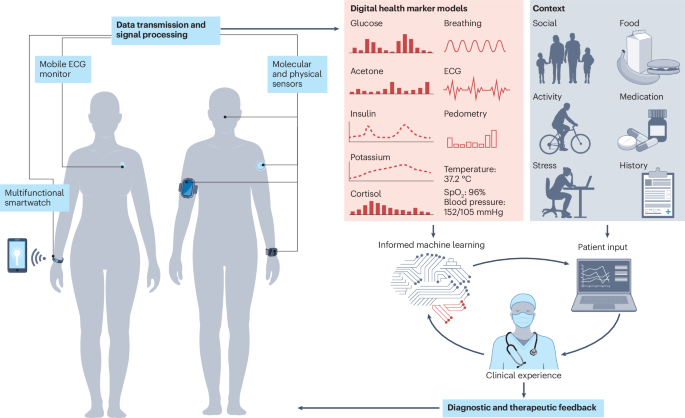
Challenges and opportunities of wearable molecular sensors in endocrinology and metabolism
Wheeler, M. J. in Hormone Assays in Biological Fluids (ed. Wheeler, M. J.) 1–6 (Humana Press, 2013).
Hirsch, I. B. in Role of Continuous Glucose Monitoring in Diabetes Treatment (ADA Clinical Compendia Series) (ed. Hirsch, I. B.) 1 (American Diabetes Association, 2018).
Goldsmith, S. J. Radioimmunoassay: review of basic principles. Semin. Nucl. Med. 5, 125–152 (1975).
Google Scholar
Shackleton, C. Clinical steroid mass spectrometry: a 45-year history culminating in HPLC–MS/MS becoming an essential tool for patient diagnosis. J. Steroid Biochem. Mol. Biol. 121, 481–490 (2010).
Google Scholar
Eisenhofer, G. et al. Reference intervals for plasma concentrations of adrenal steroids measured by LC-MS/MS: impact of gender, age, oral contraceptives, body mass index and blood pressure status. Clin. Chim. Acta 470, 115–124 (2017).
Google Scholar
Hastings, M., O’Neill, J. S. & Maywood, E. S. Circadian clocks: regulators of endocrine and metabolic rhythms. J. Endocrinol. 195, 187–198 (2007).
Google Scholar
Koop, S. & Oster, H. Eat, sleep, repeat — endocrine regulation of behavioural circadian rhythms. FEBS J. 289, 6543–6558 (2022).
Google Scholar
Tsang, A. H., Astiz, M., Friedrichs, M. & Oster, H. Endocrine regulation of circadian physiology. J. Endocrinol. 230, R1–R11 (2016).
Google Scholar
Sviridonova, M. A., Fadeyev, V. V., Sych, Y. P. & Melnichenko, G. A. Clinical significance of TSH circadian variability in patients with hypothyroidism. Endocr. Res. 38, 24–31 (2013).
Google Scholar
Sinturel, F. et al. Circadian organization of lipid landscape is perturbed in type 2 diabetic patients. Cell Rep. Med. 4, 101299 (2023).
Google Scholar
Lightman, S. L., Birnie, M. T. & Conway-Campbell, B. L. Dynamics of ACTH and cortisol secretion and implications for disease. Endocr. Rev. 41, bnaa002 (2020).
Google Scholar
Scheer, F. A., Hilton, M. F., Mantzoros, C. S. & Shea, S. A. Adverse metabolic and cardiovascular consequences of circadian misalignment. Proc. Natl Acad. Sci. USA 106, 4453–4458 (2009).
Google Scholar
Ajjan, R. A. et al. Continuous glucose monitoring for the routine care of type 2 diabetes mellitus. Nat. Rev. Endocrinol. 20, 426–440 (2024).
Google Scholar
Wang, M. et al. A wearable electrochemical biosensor for the monitoring of metabolites and nutrients. Nat. Biomed. Eng. 6, 1225–1235 (2022).
Google Scholar
Güntner, A. T. et al. Breath sensors for health monitoring. ACS Sens. 4, 268–280 (2019).
Google Scholar
Dunn, J. et al. Wearable sensors enable personalized predictions of clinical laboratory measurements. Nat. Med. 27, 1105–1112 (2021).
Google Scholar
Huang, Q., Komarzynski, S., Bolborea, M., Finkenstädt, B. & Lévi, F. A. Telemonitored human circadian temperature dynamics during daily routine. Front. Physiol. 12, 659973 (2021).
Google Scholar
Hartmeyer, S. L. et al. Multi-wearable approach for monitoring diurnal light exposure and body rhythms in nightshift workers. Acta Physiol. 241, e70069 (2025).
Google Scholar
American Diabetes Association Professional Practice Committee 7. Diabetes technology: standards of care in diabetes — 2025. Diabetes Care 48, S146–S166 (2025).
Google Scholar
Phillips, N. E. et al. The metabolic and circadian signatures of gestational diabetes in the postpartum period characterised using multiple wearable devices. Diabetologia 68, 419–432 (2025).
Google Scholar
Baker, L. B. et al. Skin-interfaced microfluidic system with personalized sweating rate and sweat chloride analytics for sports science applications. Sci. Adv. 6, eabe3929 (2020).
Google Scholar
Weber, I. C. et al. Monitoring lipolysis by sensing breath acetone down to parts-per-billion. Small Sci. 1, 2100004 (2021).
Google Scholar
Aukland, K. & Nicolaysen, G. Interstitial fluid volume: local regulatory mechanisms. Physiol. Rev. 61, 556–643 (1981).
Google Scholar
Reed, R. K. & Rubin, K. Transcapillary exchange: role and importance of the interstitial fluid pressure and the extracellular matrix. Cardiovasc. Res. 87, 211–217 (2010).
Google Scholar
Bikle, D. D. The free hormone hypothesis: when, why, and how to measure the free hormone levels to assess vitamin D, thyroid, sex hormone, and cortisol status. JBMR Plus 5, e10418 (2020).
Google Scholar
Lightman, S. L. Clinical endocrinology — time for a reset? J. Endocr. Soc. 8, bvae024 (2024).
Google Scholar
Upton, T. J. et al. High-resolution daily profiles of tissue adrenal steroids by portable automated collection. Sci. Transl. Med. 15, eadg8464 (2023).
Google Scholar
Jansson, P. A., Krogstad, A. L. & Lonnroth, P. Microdialysis measurements in skin: evidence for significant lactate release in healthy humans. Am. J. Physiol. 271, E138–E142 (1996).
Google Scholar
Jia, M. et al. Integrative bioinformatics approach identifies novel drug targets for hyperaldosteronism, with a focus on SHMT1 as a promising therapeutic candidate. Sci. Rep. 15, 1690 (2025).
Google Scholar
Fujii, N. et al. Dietary nitrate supplementation increases nitrate and nitrite concentrations in human skin interstitial fluid. Nitric Oxide 134-135, 10–16 (2023).
Google Scholar
Oharazawa, A., Maimaituxun, G., Watanabe, K., Nishiyasu, T. & Fujii, N. Metabolome analyses of skin dialysate: insights into skin interstitial fluid biomarkers. J. Dermatol. Sci. 114, 141–147 (2024).
Google Scholar
Vora, L. K. et al. Microneedle-based biosensing. Nat. Rev. Bioeng. 2, 64–81 (2024).
Google Scholar
Halder, J., Gupta, S., Kumari, R., Gupta, G. D. & Rai, V. K. Microneedle array: applications, recent advances, and clinical pertinence in transdermal drug delivery. J. Pharm. Innov. 16, 558–565 (2021).
Google Scholar
Tehrani, F. et al. An integrated wearable microneedle array for the continuous monitoring of multiple biomarkers in interstitial fluid. Nat. Biomed. Eng. 6, 1214–1224 (2022).
Google Scholar
Elias Abi-Ramia Silva, T., Kohler, S., Bartzsch, N., Beuschlein, F. & Güntner, A. 3D printing by two-photon polymerization of hollow microneedles for interstitial fluid extraction. Cell Biomater. 1, 100008 (2025).
Google Scholar
Gardeniers, H. J. G. E. et al. Silicon micromachined hollow microneedles for transdermal liquid transport. J. Microelectromech. Syst. 12, 855–862 (2003).
Google Scholar
Zhang, J. et al. Microneedle-enabled therapeutics delivery and biosensing in clinical trials. J. Controlled Rel. 360, 687–704 (2023).
Google Scholar
Harvey, C. J., LeBouf, R. F. & Stefaniak, A. B. Formulation and stability of a novel artificial human sweat under conditions of storage and use. Toxicol. Vitr. 24, 1790–1796 (2010).
Google Scholar
Gao, W. et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529, 509–514 (2016).
Google Scholar
Emaminejad, S. et al. Autonomous sweat extraction and analysis applied to cystic fibrosis and glucose monitoring using a fully integrated wearable platform. Proc. Natl Acad. Sci. USA 114, 4625–4630 (2017).
Google Scholar
Koh, A. et al. A soft, wearable microfluidic device for the capture, storage, and colorimetric sensing of sweat. Sci. Transl. Med. 8, 366ra165 (2016).
Google Scholar
Di Sant’Agnese, P. A., Darling, R. C., Perera, G. A. & Shea, E. Abnormal electrolyte composition of sweat in cystic fibrosis of the pancreas; clinical significance and relationship to the disease. Pediatrics 12, 549–563 (1953).
Google Scholar
Barben, J., Casaulta, C., Spinas, R. & Schöni, M. H. Sweat testing practice in Swiss hospitals. Swiss Med. Wkly. 137, 192–198 (2007).
Google Scholar
Bariya, M., Nyein, H. Y. Y. & Javey, A. Wearable sweat sensors. Nat. Electron. 1, 160–171 (2018).
Google Scholar
Yang, X. et al. Flexible, wearable microfluidic contact lens with capillary networks for tear diagnostics. J. Mater. Sci. 55, 9551–9561 (2020).
Google Scholar
Sempionatto, J. R. et al. Eyeglasses-based tear biosensing system: non-invasive detection of alcohol, vitamins and glucose. Biosens. Bioelectron. 137, 161–170 (2019).
Google Scholar
Drabińska, N. et al. A literature survey of all volatiles from healthy human breath and bodily fluids: the human volatilome. J. Breath. Res. 15, 034001 (2021).
Google Scholar
Puchalska, P. & Crawford, P. A. Multi-dimensional roles of ketone bodies in fuel metabolism, signaling, and therapeutics. Cell Metab. 25, 262–284 (2017).
Google Scholar
Musa-Veloso, K., Likhodii, S. S. & Cunnane, S. C. Breath acetone is a reliable indicator of ketosis in adults consuming ketogenic meals. Am. J. Clin. Nutr. 76, 65–70 (2002).
Google Scholar
Statheropoulos, M., Agapiou, A. & Georgiadou, A. Analysis of expired air of fasting male monks at Mount Athos. J. Chromatogr. Biomed. Appl. 832, 274–279 (2006).
Google Scholar
Güntner, A. T., Weber, I. C., Schon, S., Pratsinis, S. E. & Gerber, P. A. Monitoring rapid metabolic changes in health and type-1 diabetes with breath acetone sensors. Sens. Actuators B 367, 132182 (2022).
Google Scholar
Hammer, H. F. et al. European guideline on indications, performance, and clinical impact of hydrogen and methane breath tests in adult and pediatric patients: European Association for Gastroenterology, Endoscopy and Nutrition, European Society of Neurogastroenterology and Motility, and European Society for Paediatric Gastroenterology Hepatology and Nutrition consensus. United Eur. Gastroenterol. J. 10, 15–40 (2022).
Google Scholar
American Thoracic Society; European Respiratory Society ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005. Am. J. Respir. Crit. Care Med. 171, 912–930 (2005).
Google Scholar
Beauchamp, J., Herbig, J., Gutmann, R. & Hansel, A. On the use of Tedlar® bags for breath-gas sampling and analysis. J. Breath. Res. 2, 046001 (2008).
Google Scholar
King, J. et al. Isoprene and acetone concentration profiles during exercise on an ergometer. J. Breath. Res. 3, 027006 (2009).
Google Scholar
Elias Abi-Ramia Silva, T., Burisch, F. & Güntner, A. T. Gas sensing for space: health and environmental monitoring. Trends Analyt. Chem. 177, 117790 (2024).
Google Scholar
Wang, T., Pysanenko, A., Dryahina, K., Spanel, P. & Smith, D. Analysis of breath, exhaled via the mouth and nose, and the air in the oral cavity. J. Breath. Res. 2, 037013 (2008).
Google Scholar
Miekisch, W. et al. Impact of sampling procedures on the results of breath analysis. J. Breath. Res. 2, 026007 (2008).
Google Scholar
Herbig, J. & Beauchamp, J. Towards standardization in the analysis of breath gas volatiles. J. Breath. Res. 8, 037101 (2014).
Google Scholar
Yang, D. S., Ghaffari, R. & Rogers, J. A. Sweat as a diagnostic biofluid. Science 379, 760–761 (2023).
Google Scholar
Friedel, M. et al. Opportunities and challenges in the diagnostic utility of dermal interstitial fluid. Nat. Biomed. Eng. 7, 1541–1555 (2023).
Google Scholar
Cizza, G. & Rother, K. I. Cortisol binding globulin: more than just a carrier? J. Clin. Endocrinol. Metab. 97, 77–80 (2012).
Google Scholar
Hovorka, R. Closed-loop insulin delivery: from bench to clinical practice. Nat. Rev. Endocrinol. 7, 385–395 (2011).
Google Scholar
Smith, D., Wang, T., Pysanenko, A. & Spanel, P. A selected ion flow tube mass spectrometry study of ammonia in mouth- and nose-exhaled breath and in the oral cavity. Rapid Commun. Mass. Spectrom. 22, 783–789 (2008).
Google Scholar
Spanel, P., Turner, C., Wang, T., Bloor, R. & Smith, D. Generation of volatile compounds on mouth exposure to urea and sucrose: implications for exhaled breath analysis. Physiol. Meas. 27, N7–N17 (2006).
Google Scholar
Battelino, T. et al. Clinical targets for continuous glucose monitoring data interpretation: recommendations from the international consensus on time in range. Diabetes Care 42, 1593–1603 (2019).
Google Scholar
Olawade, D. B. et al. Integrating AI-driven wearable devices and biometric data into stroke risk assessment: a review of opportunities and challenges. Clin. Neurol. Neurosurg. 249, 108689 (2025).
Google Scholar
Bruderer, T. et al. On-line analysis of exhaled breath focus review. Chem. Rev. 119, 10803–10828 (2019).
Google Scholar
Elias Abi-Ramia Silva, T., Kohler, S., Bartzsch, N., Beuschlein, F. & Güntner, A. T. 3D-printed polymer hollow microneedles on microfluidic platforms for minimally invasive interstitial fluid extraction. Cell Biomater. 1, 100008 (2025).
Google Scholar
Anderson, J. C. Measuring breath acetone for monitoring fat loss: review. Obesity 23, 2327–2334 (2015).
Google Scholar
Wang, Z. et al. Microneedle patch for the ultrasensitive quantification of protein biomarkers in interstitial fluid. Nat. Biomed. Eng. 5, 64–76 (2021).
Google Scholar
Willner, I. & Zayats, M. Electronic aptamer-based sensors. Angew. Chem. Int. Ed. 46, 6408–6418 (2007).
Google Scholar
Thompson, I. A. P. et al. An antibody-based molecular switch for continuous small-molecule biosensing. Sci. Adv. 9, eadh4978 (2023).
Google Scholar
Wackerlig, J. & Lieberzeit, P. A. Molecularly imprinted polymer nanoparticles in chemical sensing — synthesis, characterisation and application. Sens. Actuators B 207, 144–157 (2015).
Google Scholar
Yeasmin, S., Wu, B., Liu, Y., Ullah, A. & Cheng, L.-J. Nano gold-doped molecularly imprinted electrochemical sensor for rapid and ultrasensitive cortisol detection. Biosens. Bioelectron. 206, 114142 (2022).
Google Scholar
Hu, X. et al. Wearable and regenerable electrochemical fabric sensing system based on molecularly imprinted polymers for real-time stress management. Adv. Funct. Mater. 34, 2312897 (2024).
Google Scholar
Stuber, A. & Nakatsuka, N. Aptamer renaissance for neurochemical biosensing. ACS Nano 18, 2552–2563 (2024).
Google Scholar
Maity, D., Guha Ray, P. & Fussenegger, M. Glucose-operated widget (GLOW) for closed-loop optogenetic glycemic control. Adv. Mater. 36, 2408537 (2024).
Google Scholar
Güntner, A. T. et al. Sniffing entrapped humans with sensor arrays. Anal. Chem. 90, 4940–4945 (2018).
Google Scholar
Xu, C., Tamaki, J., Miura, N. & Yamazoe, N. Correlation between gas sensitivity and crystallite size in porous SnO2-based sensors. Chem. Lett. 19, 441–444 (1990).
Google Scholar
Kong, J. et al. Nanotube molecular wires as chemical sensors. Science 287, 622–625 (2000).
Google Scholar
Bulemo, P. M. et al. Selectivity in chemiresistive gas sensors: strategies and challenges. Chem. Rev. 125, 4111–4183 (2025).
Google Scholar
Hibbard, T. et al. Point of care monitoring of hemodialysis patients with a breath ammonia measurement device based on printed polyaniline nanoparticle sensors. Anal. Chem. 85, 12158–12165 (2013).
Google Scholar
van den Broek, J. et al. Selective monitoring of breath isoprene by a portable detector during exercise and at rest. Sens. Actuators B 357, 131444 (2022).
Google Scholar
Güntner, A. T. et al. Guiding ketogenic diet with breath acetone sensors. Sensors 18, 3655 (2018).
Google Scholar
Chen, S. et al. Wearable flexible microfluidic sensing technologies. Nat. Rev. Bioeng. 1, 950–971 (2023).
Google Scholar
Zargartalebi, H. et al. Active-reset protein sensors enable continuous in vivo monitoring of inflammation. Science 386, 1146–1153 (2024).
Google Scholar
Frutiger, A. et al. Nonspecific binding — fundamental concepts and consequences for biosensing applications. Chem. Rev. 121, 8095–8160 (2021).
Google Scholar
Lee, J.-C. et al. Micrometer-thick and porous nanocomposite coating for electrochemical sensors with exceptional antifouling and electroconducting properties. Nat. Commun. 15, 711 (2024).
Google Scholar
Li, Y. et al. A nonfouling electrochemical biosensor for protein analysis in complex body fluids based on multifunctional peptide conjugated with PEGlyated phospholipid. ACS Sens. 9, 5596–5603 (2024).
Google Scholar
Xu, Z., Han, R., Liu, N., Gao, F. & Luo, X. Electrochemical biosensors for the detection of carcinoembryonic antigen with low fouling and high sensitivity based on copolymerized polydopamine and zwitterionic polymer. Sens. Actuators B 319, 128253 (2020).
Google Scholar
Lin, P.-H. & Li, B.-R. Antifouling strategies in advanced electrochemical sensors and biosensors. Analyst 145, 1110–1120 (2020).
Google Scholar
Güntner, A. T. et al. Noninvasive body fat burn monitoring from exhaled acetone with Si-doped WO3-sensing nanoparticles. Anal. Chem. 89, 10578–10584 (2017).
Google Scholar
Kwak, C. H. et al. Humidity-independent oxide semiconductor chemiresistors using terbium-doped SnO2 yolk-shell spheres for real-time breath analysis. ACS Appl. Mater. Interfaces 10, 18886–18894 (2018).
Google Scholar
D’Andria, M., Elias Abi-Ramia Silva, T., Consogno, E., Krumeich, F. & Güntner, A. T. Metastable CoCu2O3 nanocrystals from combustion-aerosols for molecular sensing and catalysis. Adv. Mater. 36, 2408888 (2024).
Google Scholar
D’Andria, M., Krumeich, F., Yao, Z., Wang, F. R. & Güntner, A. T. Structure-function relationship of highly reactive CuOx clusters on Co3O4 for selective formaldehyde sensing at low temperatures. Adv. Sci. 11, 2308224 (2024).
Google Scholar
Chu, T. et al. Progress and perspectives of single-atom catalysts for gas sensing. Adv. Mater. 35, 2206783 (2023).
Google Scholar
van den Broek, J., Weber, I. C., Güntner, A. & Pratsinis, S. Highly selective gas sensing enabled by filters. Mater. Horiz. 8, 661–684 (2021).
Google Scholar
Yoon, J. W. et al. A new strategy for humidity independent oxide chemiresistors: dynamic self-refreshing of In2O3 sensing surface assisted by layer-by-layer coated CeO2 nanoclusters. Small 12, 4229–4240 (2016).
Google Scholar
Abdulazeem, H., Whitelaw, S., Schauberger, G. & Klug, S. J. A systematic review of clinical health conditions predicted by machine learning diagnostic and prognostic models trained or validated using real-world primary health care data. PLoS ONE 18, e0274276 (2023).
Google Scholar
Feuerriegel, S. et al. Causal machine learning for predicting treatment outcomes. Nat. Med. 30, 958–968 (2024).
Google Scholar
Chen, H., Aebersold, H., Puhan, M. A. & Serra-Burriel, M. J. Causal machine learning methods for estimating personalised treatment effects — insights on validity from two large trials. Preprint at (2025).
Le Floch, E. et al. Identification of risk loci for primary aldosteronism in genome-wide association studies. Nat. Commun. 13, 5198 (2022).
Google Scholar
Prete, A. et al. Cardiometabolic disease burden and steroid excretion in benign adrenal tumors: a cross-sectional multicenter study. Ann. Intern. Med. 175, 325–334 (2022).
Google Scholar
Flint, S. W. & Batterham, R. L. The need to personalise approaches for the prevention and management of obesity. eClinicalMedicine 58, 101944 (2023).
Google Scholar
Funder, J. W. et al. The management of primary aldosteronism: case detection, diagnosis, and treatment: an Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 101, 1889–1916 (2016).
Google Scholar
Fleseriu, M. et al. Consensus on diagnosis and management of Cushing’s disease: a guideline update. Lancet Diabetes Endocrinol. 9, 847–875 (2021).
Google Scholar
Lenders, J. W. M. et al. Pheochromocytoma and paraganglioma: an Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 99, 1915–1942 (2014).
Google Scholar
Materson, B. J. Variability in response to antihypertensive drugs. Am. J. Med. 120, S10–S20 (2007).
Google Scholar
Ginsburg, G. S., Picard, R. W. & Friend, S. H. Key issues as wearable digital health technologies enter clinical care. N. Engl. J. Med. 390, 1118–1127 (2024).
Google Scholar
Clark, L. C. Jr. & Lyons, C. Electrode systems for continuous monitoring of cardiovascular surgery. Ann. N. Y. Acad. Sci. 102, 29–45 (1962).
Google Scholar
Wang, J. Electrochemical glucose biosensors. Chem. Rev. 108, 814–825 (2008).
Google Scholar
Cass, A. E. G. et al. Ferrocene-mediated enzyme electrode for amperometric determination of glucose. Anal. Chem. 56, 667–671 (1984).
Google Scholar
Kanter, D., Henzler, P., Jackson, A. & Diener, A. Designing the continuous glucose monitor experience: an app design process overview. J. Diabetes Sci. Technol. 18, 46–52 (2024).
Google Scholar
Howsmon, D. & Bequette, B. W. Hypo- and hyperglycemic alarms: devices and algorithms. J. Diabetes Sci. Technol. 9, 1126–1137 (2015).
Google Scholar
Advani, A. Positioning time in range in diabetes management. Diabetologia 63, 242–252 (2020).
Google Scholar
link