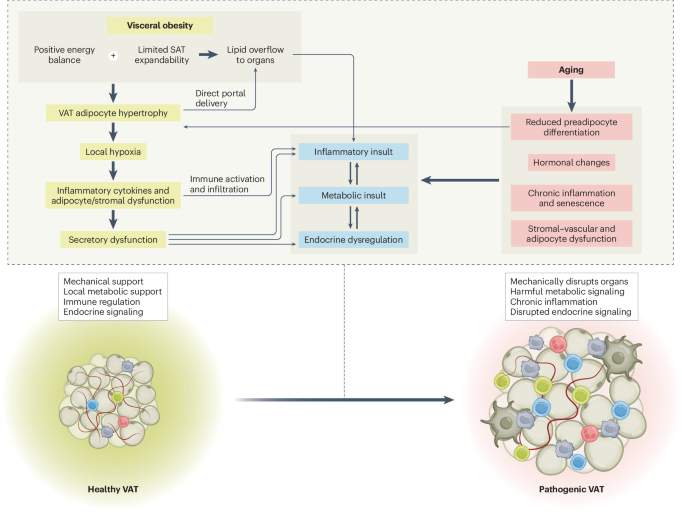
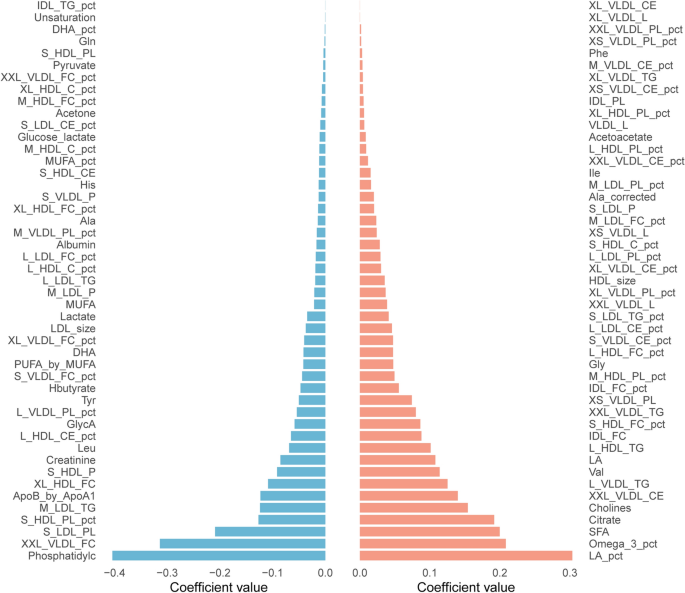
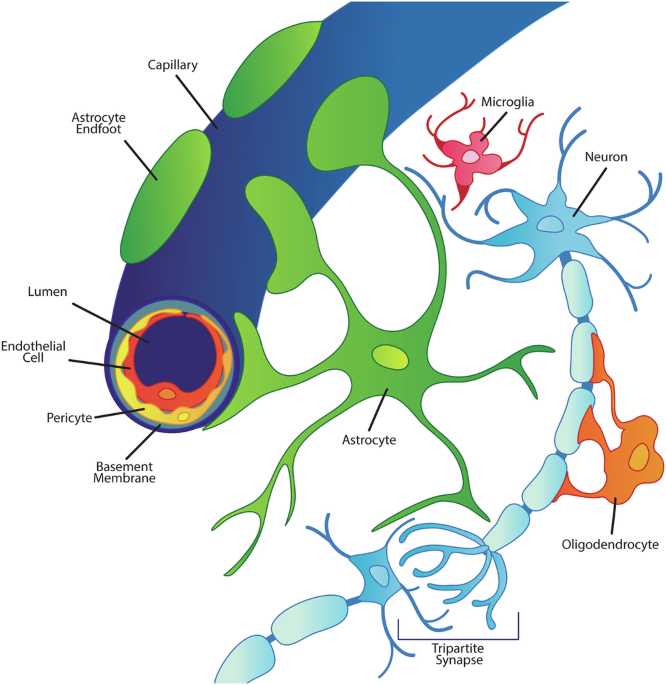
Central cytometabolic functional vascular coupling in health and disease
Iadecola, C. The neurovascular unit coming of age: a journey through neurovascular coupling in health and disease. Neuron 96, 17–42 (2017).
Google Scholar
Sokoloff, L. Local cerebral energy metabolism: its relationships to local functional activity and blood flow. Ciba Found Symp. 171–197 (1978).
Iadecola, C. et al. The neurovasculome: key roles in brain health and cognitive impairment: a scientific statement from the American Heart Association/American Stroke Association. Stroke 54, e251–e271 (2023).
Google Scholar
Attwell, D. et al. Glial and neuronal control of brain blood flow. Nature 468, 232–243 (2010).
Google Scholar
Koehler, R. C., Gebremedhin, D. & Harder, D. R. Role of astrocytes in cerebrovascular regulation. J. Appl. Physiol. 100, 307–317 (2006).
Google Scholar
Lecrux, C. & Hamel, E. The neurovascular unit in brain function and disease. Acta Physiol. 203, 47–59 (2011).
Google Scholar
Abbott, N. J., Rönnbäck, L. & Hansson, E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci. 7, 41–53 (2006).
Google Scholar
Iadecola, C. & Nedergaard, M. Glial regulation of the cerebral microvasculature. Nat. Neurosci. 10, 1369–1376 (2007).
Google Scholar
Hill, R. A. et al. Regional blood flow in the normal and ischemic brain is controlled by arteriolar smooth muscle cell contractility and not by capillary pericytes. Neuron 87, 95–110 (2015).
Google Scholar
Ross, J. M. et al. The expanding cell diversity of the brain vasculature. Front. Physiol. 11, 600767 (2020).
Google Scholar
Hartmann, D. A., Coelho-Santos, V. & Shih, A. Y. Pericyte control of blood flow across microvascular zones in the central nervous system. Annu Rev. Physiol. 84, 331–354 (2022).
Google Scholar
Cauli, B. et al. Cortical GABA interneurons in neurovascular coupling: relays for subcortical vasoactive pathways. J. Neurosci. 24, 8940–8949 (2004).
Google Scholar
Iadecola, C. Neurovascular regulation in the normal brain and in Alzheimer’s disease. Nat. Rev. Neurosci. 5, 347–360 (2004).
Google Scholar
Ding, Z. et al. Emerging roles of microglia in neuro-vascular unit: implications of microglia-neurons interactions. Front. Cell Neurosci. 15, 706025 (2021).
Google Scholar
Csaszar, E. et al. Microglia modulate blood flow, neurovascular coupling, and hypoperfusion via purinergic actions. J. Exp. Med. 219, e20211071 (2022).
Zhao, X. et al. Microglial interactions with the neurovascular system in physiology and pathology. Dev. Neurobiol. 78, 604–617 (2018).
Google Scholar
Nedergaard, M., Ransom, B. & Goldman, S. A. New roles for astrocytes: redefining the functional architecture of the brain. Trends Neurosci. 26, 523–530 (2003).
Google Scholar
Raichle, M. E. & Mintun, M. A. Brain work and brain imaging. Annu Rev. Neurosci. 29, 449–476 (2006).
Google Scholar
Drew, P. J. Neurovascular coupling: motive unknown. Trends Neurosci. 45, 809–819 (2022).
Google Scholar
Korte, N., Nortley, R. & Attwell, D. Cerebral blood flow decrease as an early pathological mechanism in Alzheimer’s disease. Acta Neuropathol. 140, 793–810 (2020).
Google Scholar
Snyder, H. M. et al. Vascular contributions to cognitive impairment and dementia including Alzheimer’s disease. Alzheimers Dement 11, 710–717 (2015).
Google Scholar
Khachaturian, Z. S. The role of calcium regulation in brain aging: reexamination of a hypothesis. Aging 1, 17–34 (1989).
Google Scholar
Shah, D. et al. Astrocyte calcium dysfunction causes early network hyperactivity in Alzheimer’s disease. Cell Rep. 40, 111280 (2022).
Google Scholar
Sompol, P. et al. Targeting astrocyte signaling alleviates cerebrovascular and synaptic function deficits in a diet-based mouse model of small cerebral vessel disease. J. Neurosci. 43, 1797–1813 (2023).
Google Scholar
Korte, N. et al. Inhibiting Ca(2+) channels in Alzheimer’s disease model mice relaxes pericytes, improves cerebral blood flow and reduces immune cell stalling and hypoxia. Nat. Neurosci. 27, 2086–2100 (2024).
Google Scholar
Andersen, J. V. et al. Hippocampal disruptions of synaptic and astrocyte metabolism are primary events of early amyloid pathology in the 5xFAD mouse model of Alzheimer’s disease. Cell Death Dis. 12, 954 (2021).
Google Scholar
Mosconi, L. et al. FDG-PET changes in brain glucose metabolism from normal cognition to pathologically verified Alzheimer’s disease. Eur. J. Nucl. Med Mol. Imaging 36, 811–822 (2009).
Google Scholar
Brooks, W. M. et al. Gene expression profiles of metabolic enzyme transcripts in Alzheimer’s disease. Brain Res. 1127, 127–135 (2007).
Google Scholar
Gannon, M. & Wang, Q. Complex noradrenergic dysfunction in Alzheimer’s disease: Low norepinephrine input is not always to blame. Brain Res. 1702, 12–16 (2019).
Google Scholar
Frolich, L. et al. A disturbance in the neuronal insulin receptor signal transduction in sporadic Alzheimer’s disease. Ann. N. Y Acad. Sci. 893, 290–293 (1999).
Google Scholar
Pancani, T. et al. Distinct modulation of voltage-gated and ligand-gated Ca2+ currents by PPAR-gamma agonists in cultured hippocampal neurons. J. Neurochem. 109, 1800–1811 (2009).
Google Scholar
Maimaiti, S. et al. Novel calcium-related targets of insulin in hippocampal neurons. Neuroscience 364, 130–142 (2017).
Google Scholar
Lin, R. L. et al. Sensitivity of the S1 neuronal calcium network to insulin and Bay-K 8644 in vivo: relationship to gait, motivation, and aging processes. Aging Cell 21, e13661 (2022).
Google Scholar
Foo, L. C. et al. Development of a method for the purification and culture of rodent astrocytes. Neuron 71, 799–811 (2011).
Google Scholar
Renden, R. B. et al. Modulatory effects of noradrenergic and serotonergic signaling pathway on neurovascular coupling. Commun. Biol. 7, 287 (2024).
Google Scholar
Sofroniew, M. V. & Vinters, H. V. Astrocytes: biology and pathology. Acta Neuropathol. 119, 7–35 (2010).
Google Scholar
Cai, W. et al. Insulin regulates astrocyte gliotransmission and modulates behavior. J. Clin. Investig. 128, 2914–2926 (2018).
Google Scholar
Pellerin, L. & Magistretti, P. J. Glutamate uptake into astrocytes stimulates aerobic glycolysis—a mechanism coupling neuronal-activity to glucose-utilization. Proc. Natl. Acad. Sci. USA 91, 10625–10629 (1994).
Google Scholar
Yellen, G. Fueling thought: Management of glycolysis and oxidative phosphorylation in neuronal metabolism. J. Cell Biol. 217, 2235–2246 (2018).
Google Scholar
Frazier, H. N. et al. Elevating insulin signaling using a constitutively active insulin receptor increases glucose metabolism and expression of GLUT3 in hippocampal neurons. Front. Neurosci. 14, 668 (2020).
Google Scholar
Pancani, T. et al. Imaging of a glucose analog, calcium and NADH in neurons and astrocytes: Dynamic responses to depolarization and sensitivity to pioglitazone. Cell Calcium 50, 548–558 (2011).
Google Scholar
Diaz-Garcia, C. M. et al. Neuronal Stimulation Triggers Neuronal Glycolysis and Not Lactate Uptake. Cell Metab. 26, 361–374 e4 (2017).
Google Scholar
Bak, L. K. et al. Neuronal glucose but not lactate utilization is positively correlated with NMDA-induced neurotransmission and fluctuations in cytosolic Ca2+ levels. J. Neurochem. 109, 87–93 (2009).
Google Scholar
Lundgaard, I. et al. Direct neuronal glucose uptake heralds activity-dependent increases in cerebral metabolism. Nat. Commun. 6, 6807 (2015).
Google Scholar
Dienel, G. A. Brain lactate metabolism: the discoveries and the controversies. J. Cereb. Blood Flow. Metab. 32, 1107–1138 (2012).
Google Scholar
Hertz, L., Peng, L. & Dienel, G. A. Energy metabolism in astrocytes: high rate of oxidative metabolism and spatiotemporal dependence on glycolysis/glycogenolysis. J. Cereb. Blood Flow. Metab. 27, 219–249 (2007).
Google Scholar
Verkhratsky, A. et al. Astrocytes in human central nervous system diseases: a frontier for new therapies. Signal Transduct. Target Ther. 8, 396 (2023).
Google Scholar
Takata, N. et al. Astrocyte calcium signaling transforms cholinergic modulation to cortical plasticity in vivo. J. Neurosci. 31, 18155–18165 (2011).
Google Scholar
Haydon, P. G. & Carmignoto, G. Astrocyte control of synaptic transmission and neurovascular coupling. Physiol. Rev. 86, 1009–1031 (2006).
Google Scholar
Tran, C. H. T., Peringod, G. & Gordon, G. R. Astrocytes integrate behavioral state and vascular signals during functional hyperemia. Neuron 100, 1133–1148 e3 (2018).
Google Scholar
Del Franco, A. P., Chiang, P. P. & Newman, E. A. Dilation of cortical capillaries is not related to astrocyte calcium signaling. Glia 70, 508–521 (2022).
Google Scholar
Institoris, Á, Rosenegger, D. G. & Gordon, G. R. Arteriole dilation to synaptic activation that is sub-threshold to astrocyte endfoot Ca2+ transients. J. Cereb. Blood Flow. Metab. 35, 1411–1415 (2015).
Google Scholar
Rosenegger, D. G. et al. Tonic local brain blood flow control by astrocytes independent of phasic neurovascular coupling. J. Neurosci. 35, 13463–13474 (2015).
Google Scholar
Nagele, R. G. et al. Contribution of glial cells to the development of amyloid plaques in Alzheimer’s disease. Neurobiol. Aging 25, 663–674 (2004).
Google Scholar
Norris, C. M. et al. Calcineurin triggers reactive/inflammatory processes in astrocytes and is upregulated in aging and Alzheimer’s models. J. Neurosci. 25, 4649–4658 (2005).
Google Scholar
Huffels, C. F. M. et al. Calcium signaling in individual APP/PS1 mouse dentate gyrus astrocytes increases ex vivo with Abeta pathology and age without affecting astrocyte network activity. J. Neurosci. Res. 100, 1281–1295 (2022).
Google Scholar
Mitroshina, E. V. et al. Novel algorithm of network calcium dynamics analysis for studying the role of astrocytes in neuronal activity in Alzheimer’s disease models. Int. J. Mol. Sci. 23, 15928 (2022).
Verkhratsky, A. et al. Astrocytes in Alzheimer’s disease. Neurotherapeutics 7, 399–412 (2010).
Google Scholar
Olabarria, M. et al. Concomitant astroglial atrophy and astrogliosis in a triple transgenic animal model of Alzheimer’s disease. Glia 58, 831–838 (2010).
Google Scholar
Zhukov, O. et al. Preserved blood-brain barrier and neurovascular coupling in female 5xFAD model of Alzheimer’s disease. Front. Aging Neurosci. 15, 1089005 (2023).
Google Scholar
Krueger, M. & Bechmann, I. CNS pericytes: concepts, misconceptions, and a way out. Glia 58, 1–10 (2010).
Google Scholar
Hall, C. N. et al. Capillary pericytes regulate cerebral blood flow in health and disease. Nature 508, 55–60 (2014).
Google Scholar
Meza-Resillas, J. et al. Systemic nimodipine affects pericyte calcium signaling, resting hemodynamics and neurovascular coupling in healthy mouse brain. Neurotherapeutics. 22, e00614 (2025).
Nikolakopoulou, A. M. et al. Pericyte loss leads to circulatory failure and pleiotrophin depletion causing neuron loss. Nat. Neurosci. 22, 1089–1098 (2019).
Google Scholar
Stobart, J. L. et al. Altered hemodynamics and vascular reactivity in a mouse model with severe pericyte deficiency. J. Cereb. Blood Flow. Metab. 43, 763–777 (2023).
Google Scholar
Kisler, K. et al. Pericyte degeneration leads to neurovascular uncoupling and limits oxygen supply to brain. Nat. Neurosci. 20, 406–416 (2017).
Google Scholar
Li, P. & Fan, H. Pericyte loss in diseases. Cells. 12 (2023).
Costa, E. D. et al. Neuronal nitric oxide synthase in vascular physiology and diseases. Front Physiol. 7, 206 (2016).
Google Scholar
Irikura, K. et al. Cerebrovascular alterations in mice lacking neuronal nitric oxide synthase gene expression. Proc. Natl. Acad. Sci. USA 92, 6823–6827 (1995).
Google Scholar
Hosford, P. S. & Gourine, A. V. What is the key mediator of the neurovascular coupling response?. Neurosci. Biobehav. Rev. 96, 174–181 (2019).
Google Scholar
Meng, W. et al. ACh dilates pial arterioles in endothelial and neuronal NOS knockout mice by NO-dependent mechanisms. Am. J. Physiol. 271, H1145–H1150 (1996).
Google Scholar
Prast, H. et al. Nitric oxide-induced release of acetylcholine in the nucleus accumbens: role of cyclic GMP, glutamate, and GABA. J. Neurochem. 71, 266–273 (1998).
Google Scholar
Guevara-Guzman, R., Emson, P. C. & Kendrick, K. M. Modulation of in vivo striatal transmitter release by nitric oxide and cyclic GMP. J. Neurochem. 62, 807–810 (1994).
Google Scholar
Elhusseiny, A. & Hamel, E. Muscarinic—but not nicotinic—acetylcholine receptors mediate a nitric oxide-dependent dilation in brain cortical arterioles: a possible role for the M5 receptor subtype. J. Cereb. Blood Flow. Metab. 20, 298–305 (2000).
Google Scholar
Zhou, M. et al. Blood pressure partially mediated the association of insulin resistance and cerebral small vessel disease: a community-based study. J. Am. Heart Assoc. 13, e031723 (2024).
Google Scholar
Yamada, M. et al. Cholinergic dilation of cerebral blood vessels is abolished in M(5) muscarinic acetylcholine receptor knockout mice. Proc. Natl. Acad. Sci. USA 98, 14096–14101 (2001).
Google Scholar
Lacroix, A. et al. COX-2-derived prostaglandin E2 produced by pyramidal neurons contributes to neurovascular coupling in the rodent cerebral cortex. J. Neurosci. 35, 11791–11810 (2015).
Google Scholar
He, Y. et al. Ultra-slow single-vessel BOLD and CBV-based fMRI spatiotemporal dynamics and their correlation with neuronal intracellular calcium signals. Neuron 97, 925–939 e5 (2018).
Google Scholar
Rosenblum, W. I. Inhibition of barium-induced constriction of cerebral surface arterioles by blockers of calcium channels. Blood Vessels 22, 139–144 (1985).
Google Scholar
Rosenblum, W. I., McDonald, M. & Wormley, B. Calcium ionophore and acetylcholine dilate arterioles on the mouse brain by different mechanisms. Stroke 20, 1391–1395 (1989).
Google Scholar
Rosenblum, W. I. Cerebral microcirculation: a review emphasizing the interrelationship of local blood flow and neuronal function. Angiology 16, 485–507 (1965).
Google Scholar
Skinhoj, E. Regulation of cerebral blood flow as a single function of the interstitial pH in the brain. A hypothesis. Acta Neurol. Scand. 42, 604–607 (1966).
Google Scholar
Caldwell, H. G. Blunted cerebrovascular CO(2) reactivity to satisfy the hungry heat stressed brain. J. Physiol. 599, 2513–2515 (2021).
Google Scholar
Roche, M. et al. In vivo imaging with a water immersion objective affects brain temperature, blood flow and oxygenation. Elife. 8, e47324 (2019).
Iadecola, C. & Gottesman, R. F. Cerebrovascular alterations in Alzheimer disease. Circ. Res 123, 406–408 (2018).
Google Scholar
Rosenblum, W. I. Contractile response of pial arterioles to norepinephrine. Effects in the mouse. Arch. Neurol. 31, 197–199 (1974).
Google Scholar
Lauritzen, M. Reading vascular changes in brain imaging: is dendritic calcium the key. Nat. Rev. Neurosci. 6, 77–85 (2005).
Google Scholar
Agulhon, C. et al. Calcium Signaling and Gliotransmission in Normal vs. Reactive Astrocytes. Front Pharm. 3, 139 (2012).
Google Scholar
Lalo, U. & Pankratov, Y. Astrocyte ryanodine receptors facilitate gliotransmission and astroglial modulation of synaptic plasticity. Front Cell Neurosci. 18, 1382010 (2024).
Google Scholar
Mishra, A. et al. Astrocytes mediate neurovascular signaling to capillary pericytes but not to arterioles. Nat. Neurosci. 19, 1619–1627 (2016).
Google Scholar
Stella, N. et al. Glutamate induces the release of arachidonic acid by interacting with an atypical metabotropic receptor present on mouse brain astrocytes. Ren. Physiol. Biochem. 17, 153–156 (1994).
Google Scholar
Metea, M. R. & Newman, E. A. Glial cells dilate and constrict blood vessels: a mechanism of neurovascular coupling. J. Neurosci. 26, 2862–2870 (2006).
Google Scholar
MacVicar, B. A. & Newman, E. A. Astrocyte regulation of blood flow in the brain. Cold Spring Harb. Perspect. Biol. 7, a020388 (2015).
Shi, Y. et al. Interaction of mechanisms involving epoxyeicosatrienoic acids, adenosine receptors, and metabotropic glutamate receptors in neurovascular coupling in rat whisker barrel cortex. J. Cereb. Blood Flow. Metab. 28, 111–125 (2007).
Google Scholar
Mulligan, S. J. & MacVicar, B. A. Calcium transients in astrocyte endfeet cause cerebrovascular constrictions. Nature 431, 195–199 (2004).
Google Scholar
Takano, T. et al. Astrocyte-mediated control of cerebral blood flow. Nat. Neurosci. 9, 260–267 (2006).
Google Scholar
Zonta, M. et al. Neuron-to-astrocyte signaling is central to the dynamic control of brain microcirculation. Nat. Neurosci. 6, 43–50 (2003).
Google Scholar
Bonder, D. E. & McCarthy, K. D. Astrocytic Gq-GPCR-linked IP3R-dependent Ca2+ signaling does not mediate neurovascular coupling in mouse visual cortex in vivo. J. Neurosci. 34, 13139–13150 (2014).
Google Scholar
Nizar, K. et al. In vivo stimulus-induced vasodilation occurs without IP3 receptor activation and may precede astrocytic calcium increase. J. Neurosci. 33, 8411–8422 (2013).
Google Scholar
He, L., Linden, D. J. & Sapirstein, A. Astrocyte inositol triphosphate receptor type 2 and cytosolic phospholipase A2 alpha regulate arteriole responses in mouse neocortical brain slices. PLoS One 7, e42194 (2012).
Google Scholar
De Leo, C., Eftimiadi, C. & Schito, G. C. Rapid disappearance from the intestinal tract of bacteria resistant to rifaximin. Drugs Exp. Clin. Res 12, 979–981 (1986).
Google Scholar
Ozawa, K. et al. Astrocytic GPCR-induced Ca(2+) signaling is not causally related to local cerebral blood flow changes. Int. J. Mol. Sci. 24 (2023).
Srinivasan, R. et al. Ca(2+) signaling in astrocytes from Ip3r2(-/-) mice in brain slices and during startle responses in vivo. Nat. Neurosci. 18, 708–717 (2015).
Google Scholar
Stobart, J. L. et al. Cortical circuit activity evokes rapid astrocyte calcium signals on a similar timescale to neurons. Neuron 98, 726–735 e4 (2018).
Google Scholar
Paulson, O. B. & Newman, E. A. Does the release of potassium from astrocyte endfeet regulate cerebral blood flow?. Science 237, 896–898 (1987).
Google Scholar
Gordon, G. R. et al. Brain metabolism dictates the polarity of astrocyte control over arterioles. Nature 456, 745–749 (2008).
Google Scholar
Oe, Y. et al. Distinct temporal integration of noradrenaline signaling by astrocytic second messengers during vigilance. Nat. Commun. 11, 471 (2020).
Google Scholar
Reitman, M. E. et al. Norepinephrine links astrocytic activity to regulation of cortical state. Nat. Neurosci. 26, 579–593 (2023).
Google Scholar
Wang, Z. H. et al. Therapeutic effects of astrocytes expressing both tyrosine hydroxylase and brain-derived neurotrophic factor on a rat model of Parkinson’s disease. Neuroscience 113, 629–640 (2002).
Google Scholar
Li, X. M. et al. Gene expression of aromatic L-amino acid decarboxylase in cultured rat glial cells. J. Neurochem 59, 1172–1175 (1992).
Google Scholar
Vardjan, N., Verkhratsky, A. & Zorec, R. Pathologic potential of astrocytic vesicle traffic: new targets to treat neurologic diseases?. Cell Transplant. 24, 599–612 (2015).
Google Scholar
Filosa, J. A., Bonev, A. D. & Nelson, M. T. Calcium dynamics in cortical astrocytes and arterioles during neurovascular coupling. Circ. Res. 95, e73–e81 (2004).
Google Scholar
Haidey, J. N. et al. Astrocytes regulate ultra-slow arteriole oscillations via stretch-mediated TRPV4-COX-1 feedback. Cell Rep. 36, 109405 (2021).
Google Scholar
Institoris, A. et al. Astrocytes amplify neurovascular coupling to sustained activation of neocortex in awake mice. Nat. Commun. 13, 7872 (2022).
Google Scholar
Weiss, B. E. et al. Disrupted calcium dynamics in reactive astrocytes occur with endfeet-arteriole decoupling in an amyloid mouse model of Alzheimer’s disease. bioRxiv. p. 2025.01.24.634584 (2025).
Lim, D. et al. Amyloid beta deregulates astroglial mGluR5-mediated calcium signaling via calcineurin and Nf-kB. (1098-1136 (Electronic)).
Ronco, V. et al. Differential deregulation of astrocytic calcium signalling by amyloid-β, TNFα, IL-1β and LPS. Cell Calcium, 55, 219–229 (2014).
Sanchez-Mico, M. V. et al. Soluble Abeta produces an increase in cytosolic and mitochondrial calcium in astrocytes in vivo, in part by direct effect in astroglial cells. Alzheimer’s. Dement. 19, e077883 (2023).
Google Scholar
Kook, S. Y. et al. Aβ₁₋₄₂-RAGE interaction disrupts tight junctions of the blood-brain barrier via Ca²⁺-calcineurin signaling. J. Nneurosci. 32, 8845–8854 (2012).
Grutzendler, J. & Nedergaard, M. Cellular control of brain capillary blood flow: in vivo imaging veritas. Trends Neurosci. 42, 528–536 (2019).
Google Scholar
Wei, H. S. et al. Erythrocytes are oxygen-sensing regulators of the cerebral microcirculation. Neuron 91, 851–862 (2016).
Google Scholar
Berthiaume, A. A. et al. Pericyte remodeling is deficient in the aged brain and contributes to impaired capillary flow and structure. Nat. Commun. 13, 5912 (2022).
Google Scholar
Nielson, C. D. & Shih, A. Y. In vivo single cell optical ablation of brain pericytes. Front. Neurosci. 16, 900761 (2022).
Google Scholar
Coelho-Santos, V. & Shih, A. Y. Pericytes: unsung heroes in myelin repair after neonatal brain hypoxia. Neuron 112, 2081–2083 (2024).
Google Scholar
Balbi, M. et al. Dysfunction of mouse cerebral arteries during early aging. J. Cereb. Blood Flow. Metab. 35, 1445–1453 (2015).
Google Scholar
Rucker, H. K., Wynder, H. J. & Thomas, W. E. Cellular mechanisms of CNS pericytes. Brain Res. Bull. 51, 363–369 (2000).
Google Scholar
Sweeney, M. D., Ayyadurai, S. & Zlokovic, B. V. Pericytes of the neurovascular unit: key functions and signaling pathways. Nat. Neurosci. 19, 771–783 (2016).
Google Scholar
Markhotina, N., Liu, G. J. & Martin, D. K. Contractility of retinal pericytes grown on silicone elastomer substrates is through a protein kinase A-mediated intracellular pathway in response to vasoactive peptides. IET Nanobiotechnol. 1, 44–51 (2007).
Google Scholar
Peppiatt, C. M. et al. Bidirectional control of CNS capillary diameter by pericytes. Nature 443, 700–704 (2006).
Google Scholar
Korte, N. et al. Noradrenaline released from locus coeruleus axons contracts cerebral capillary pericytes via alpha(2) adrenergic receptors. J. Cereb. Blood Flow. Metab. 43, 1142–1152 (2023).
Google Scholar
Horlyck, S. et al. ATP induces contraction of cultured brain capillary pericytes via activation of P2Y-type purinergic receptors. Am. J. Physiol. Heart Circ. Physiol. 320, H699–H712 (2021).
Google Scholar
Gluck, C. et al. Distinct signatures of calcium activity in brain mural cells. Elife. 10, e70591 (2021).
Hariharan, A. et al. Brain capillary pericytes are metabolic sentinels that control blood flow through a K(ATP) channel-dependent energy switch. Cell Rep. 41, 111872 (2022).
Google Scholar
Gonzales, A. L. et al. Contractile pericytes determine the direction of blood flow at capillary junctions. Proc. Natl. Acad. Sci. USA 117, 27022–27033 (2020).
Google Scholar
Pearson-Leary, J. et al. Insulin modulates hippocampally-mediated spatial working memory via glucose transporter-4. Behav. Brain Res. 338, 32–39 (2018).
Google Scholar
Garcia-Caceres, C. et al. Astrocytic insulin signaling couples brain glucose uptake with nutrient availability. Cell 166, 867–880 (2016).
Google Scholar
Boyd, F. T. Jr. et al. Insulin receptors and insulin modulation of norepinephrine uptake in neuronal cultures from rat brain. J. Biol. Chem. 260, 15880–15884 (1985).
Google Scholar
Yorek, M. A., Dunlap, J. A. & Ginsberg, B. H. Amino acid and putative neurotransmitter transport in human Y79 retinoblastoma cells. Effect of insulin and insulin-like growth factor. J. Biol. Chem. 262, 10986–10993 (1987).
Google Scholar
Heidenreich, K. A. et al. Insulin stimulates the activity of a novel protein kinase C, PKC-epsilon, in cultured fetal chick neurons. J. Biol. Chem. 265, 15076–15082 (1990).
Google Scholar
Havrankova, J., Roth, J. & Brownstein, M. Insulin receptors are widely distributed in the central nervous system of the rat. Nature 272, 827–829 (1978).
Google Scholar
Hopkins, D. F. & Williams, G. Insulin receptors are widely distributed in human brain and bind human and porcine insulin with equal affinity. Diabet. Med. 14, 1044–1050 (1997).
Google Scholar
Clarke, D. W. et al. Insulin binds to specific receptors and stimulates 2-deoxy-D-glucose uptake in cultured glial cells from rat brain. J. Biol. Chem. 259, 11672–11675 (1984).
Google Scholar
Kum, W. et al. Insulin binding and effects on pyrimidine nucleoside uptake and incorporation in cultured mouse astrocytes. J. Neurochem. 49, 1293–1300 (1987).
Google Scholar
Bar, R. S., Hoak, J. C. & Peacock, M. L. Insulin receptors in human endothelial cells: identification and characterization. J. Clin. Endocrinol. Metab. 47, 699–702 (1978).
Google Scholar
Garwood, C. J. et al. Insulin and IGF1 signalling pathways in human astrocytes in vitro and in vivo; characterisation, subcellular localisation and modulation of the receptors. Mol. Brain 8, 51 (2015).
Google Scholar
Spencer, B. et al. Identification of insulin receptor splice variant B in neurons by in situ detection in human brain samples. Sci. Rep. 8, 4070 (2018).
Google Scholar
Andersen, M. et al. IGF1 and IGF2 specificities to the two insulin receptor isoforms are determined by insulin receptor amino acid 718. Plos One 12, e0178885 (2017).
Google Scholar
Belfiore, A. et al. Insulin receptor isoforms in physiology and disease: an updated view. Endocr. Rev. 38, 379–431 (2017).
Google Scholar
Malakar, P. et al. Insulin receptor alternative splicing is regulated by insulin signaling and modulates beta cell survival. Sci. Rep. 6, 31222 (2016).
Google Scholar
Vogt, B. et al. The two isotypes of the human insulin receptor (HIR-A and HIR-B) follow different internalization kinetics. Biochem. Biophys. Res. Commun. 177, 1013–1018 (1991).
Google Scholar
Yamaguchi, Y. et al. Functional properties of two naturally occurring isoforms of the human insulin receptor in Chinese hamster ovary cells. Endocrinology 129, 2058–2066 (1991).
Google Scholar
Belfiore, A. et al. Insulin receptor isoforms and insulin receptor/insulin-like growth factor receptor hybrids in physiology and disease. Endocr. Rev. 30, 586–623 (2009).
Google Scholar
Frasca, F. et al. Insulin receptor isoform A, a newly recognized, high-affinity insulin-like growth factor II receptor in fetal and cancer cells. Mol. Cell. Biol. 19, 3278–3288 (1999).
Google Scholar
Vella, V. et al. Novel mechanisms of tumor promotion by the insulin receptor isoform A in triple-negative breast cancer cells. Cells. 10 (2021).
White, M. F. & Kahn, C. R. The insulin signaling system. J. Biol. Chem. 269, 1-4 (1994).
Arnold, S. E. et al. Brain insulin resistance in type 2 diabetes and Alzheimer disease: concepts and conundrums. Nat. Rev. Neurol. 14, 168–181 (2018).
Google Scholar
Frolich, L. et al. Brain insulin and insulin receptors in aging and sporadic Alzheimer’s disease. J. Neural Transm. 105, 423–438 (1998).
Google Scholar
Stanley, M., Macauley, S. L. & Holtzman, D. M. Changes in insulin and insulin signaling in Alzheimer’s disease: cause or consequence?. J. Exp. Med. 213, 1375–1385 (2016).
Google Scholar
Pessin, J. E. & Saltiel, A. R. Signaling pathways in insulin action: molecular targets of insulin resistance. J. Clin. Investig. 106, 165–169 (2000).
Google Scholar
Frazier, H. N. et al. Broadening the definition of brain insulin resistance in aging and Alzheimer’s disease. Exp. Neurol. 313, 79–87 (2019).
Google Scholar
Banks, W. A., Owen, J. B. & Erickson, M. A. Insulin in the brain: there and back again. Pharm. Ther. 136, 82–93 (2012).
Google Scholar
Milstein, J. L. & Ferris, H. A. The brain as an insulin-sensitive metabolic organ. Mol. Metab. 52, 101234 (2021).
Google Scholar
de la Monte, S. M. Brain insulin resistance and deficiency as therapeutic targets in Alzheimer’s disease. Curr. Alzheimer Res. 9, 35–66 (2012).
Google Scholar
Razani, E. et al. The PI3K/Akt signaling axis in Alzheimer’s disease: a valuable target to stimulate or suppress?. Cell Stress Chaperones 26, 871–887 (2021).
Google Scholar
Freude, S., Schilbach, K. & Schubert, M. The role of IGF-1 receptor and insulin receptor signaling for the pathogenesis of Alzheimer’s disease: from model organisms to human disease. Curr. Alzheimer Res. 6, 213–223 (2009).
Google Scholar
Talbot, K. et al. Demonstrated brain insulin resistance in Alzheimer’s disease patients is associated with IGF-1 resistance, IRS-1 dysregulation, and cognitive decline. J. Clin. Investig. 122, 1316–1338 (2012).
Google Scholar
Tian, Y., Jing, G. & Zhang, M. Insulin-degrading enzyme: roles and pathways in ameliorating cognitive impairment associated with Alzheimer’s disease and diabetes. Ageing Res. Rev. 90, 101999 (2023).
Google Scholar
Soleimanzad, H. et al. Obesity in midlife hampers resting and sensory-evoked cerebral blood flow in mice. Obesity 29, 150–158 (2021).
Google Scholar
Li, W. et al. Early effects of high-fat diet on neurovascular function and focal ischemic brain injury. Am. J. Physiol. Regul. Integr. Comp. Physiol. 304, R1001–R1008 (2013).
Google Scholar
Craft, S. et al. Memory improvement following induced hyperinsulinemia in Alzheimer’s disease. Neurobiol. Aging 17, 123–130 (1996).
Google Scholar
Reger, M. A. et al. Effects of intranasal insulin on cognition in memory-impaired older adults: modulation by APOE genotype. Neurobiol. Aging 27, 451–458 (2006).
Google Scholar
Benedict, C. et al. Intranasal insulin improves memory in humans: superiority of insulin aspart. Neuropsychopharmacology 32, 239–243 (2007).
Google Scholar
Hallschmid, M. et al. Obese men respond to cognitive but not to catabolic brain insulin signaling. Int J. Obes. 32, 275–282 (2008).
Google Scholar
Reger, M. A. et al. Intranasal insulin administration dose-dependently modulates verbal memory and plasma amyloid-beta in memory-impaired older adults. J. Alzheimers Dis. 13, 323–331 (2008).
Google Scholar
Maimaiti, S. et al. Intranasal insulin improves age-related cognitive deficits and reverses electrophysiological correlates of brain aging. J. Gerontol. A Biol. Sci. Med. Sci. 71, 30–39 (2016).
Google Scholar
Pancani, T. et al. Effect of high-fat diet on metabolic indices, cognition, and neuronal physiology in aging F344 rats. Neurobiol. Aging 34, 1977–1987 (2013).
Google Scholar
Anderson, K. L. et al. Impact of single or repeated dose intranasal zinc-free insulin in young and aged F344 rats on cognition, signaling, and brain metabolism. J. Gerontol. A Biol. Sci. Med. Sci. 72, 189–197 (2017).
Google Scholar
Taib, B. et al. Insulin acts on astrocytes to shift their substrate preference to fatty acids. iScience 28, 111642 (2025).
Google Scholar
Hughes, T. M. & Craft, S. The role of insulin in the vascular contributions to age-related dementia. Biochim. Biophys. Acta 1862, 983–991 (2016).
Google Scholar
Potenza, M. A. et al. Treatment of spontaneously hypertensive rats with rosiglitazone and/or enalapril restores balance between vasodilator and vasoconstrictor actions of insulin with simultaneous improvement in hypertension and insulin resistance. Diabetes 55, 3594–3603 (2006).
Google Scholar
Cabou, C. et al. Central insulin regulates heart rate and arterial blood flow: an endothelial nitric oxide synthase-dependent mechanism altered during. Diab. Diab. 56, 2872–2877 (2007).
Google Scholar
Konishi, M. et al. Endothelial insulin receptors differentially control insulin signaling kinetics in peripheral tissues and brain of mice. Proc. Natl. Acad. Sci. USA 114, E8478-E8487 (2017).
Google Scholar
Fernandez, A. M. et al. Insulin regulates neurovascular coupling through astrocytes. Proc. Natl. Acad. Sci. USA 119, e2204527119 (2022).
Google Scholar
Kullmann, S. et al. Insulin action in the human brain: evidence from neuroimaging studies. J. Neuroendocrinol. 27, 419–423 (2015).
Google Scholar
Akintola, A. A. et al. Effect of intranasally administered insulin on cerebral blood flow and perfusion; a randomized experiment in young and older adults. Aging 9, 790–802 (2017).
Google Scholar
Safar, M. E. et al. Hypertension and vascular dynamics in men and women with metabolic syndrome. J. Am. Coll. Cardiol. 61, 12–19 (2013).
Google Scholar
Ausk, K. J., Boyko, E. J. & Ioannou, G. N. Insulin resistance predicts mortality in nondiabetic individuals in the U.S. Diab. Care 33, 1179–1185 (2010).
Google Scholar
Leclerc, M. et al. Cerebrovascular insulin receptors are defective in Alzheimer’s disease. Brain 146, 75–90 (2023).
Google Scholar
Case, S. L. et al. Falling short: the contribution of central insulin receptors to gait dysregulation in brain aging. Biomedicines. 10 (2022).
Chaigneau, E. et al. The relationship between blood flow and neuronal activity in the rodent olfactory bulb. J. Neurosci. 27, 6452–6460 (2007).
Google Scholar
Winship, I. R., Plaa, N. & Murphy, T. H. Rapid astrocyte calcium signals correlate with neuronal activity and onset of the hemodynamic response in vivo. J. Neurosci. 27, 6268–6272 (2007).
Google Scholar
Filosa, J. A. et al. Local potassium signaling couples neuronal activity to vasodilation in the brain. Nat. Neurosci. 9, 1397–1403 (2006).
Google Scholar
Paukert, M. et al. Norepinephrine controls astroglial responsiveness to local circuit activity. Neuron 82, 1263–1270 (2014).
Google Scholar
Bojarskaite, L. et al. Astrocytic Ca(2+) signaling is reduced during sleep and is involved in the regulation of slow wave sleep. Nat. Commun. 11, 3240 (2020).
Google Scholar
Ding, F. et al. alpha1-Adrenergic receptors mediate coordinated Ca2+ signaling of cortical astrocytes in awake, behaving mice. Cell Calcium 54, 387–394 (2013).
Google Scholar
Gordon, G. R. & Bains, J. S. Noradrenaline triggers multivesicular release at glutamatergic synapses in the hypothalamus. J. Neurosci. 25, 11385–11395 (2005).
Google Scholar
link