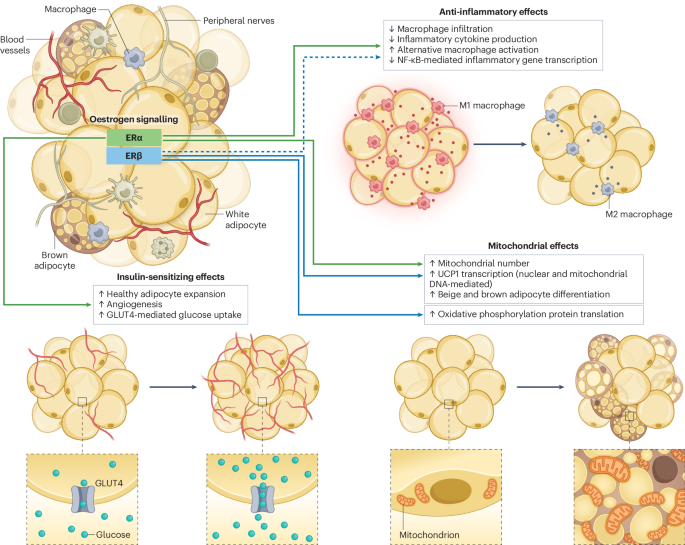
Health of adipose tissue: oestrogen matters
Amengual-Cladera, E., Llado, I., Gianotti, M. & Proenza, A. M. Sex differences in the effect of high-fat diet feeding on rat white adipose tissue mitochondrial function and insulin sensitivity. Metabolism 61, 1108–1117 (2012).
Google Scholar
Shen, M., Kumar, S. P. & Shi, H. Estradiol regulates insulin signaling and inflammation in adipose tissue. Horm. Mol. Biol. Clin. Investig. 17, 99–107 (2014).
Google Scholar
Brand, J. S. et al. Age at menopause, reproductive life span, and type 2 diabetes risk: results from the EPIC-InterAct study. Diabetes Care 36, 1012–1019 (2012).
Google Scholar
Zaydun, G. et al. Menopause is an independent factor augmenting the age-related increase in arterial stiffness in the early postmenopausal phase. Atherosclerosis 184, 137–142 (2006).
Google Scholar
Varlamov, O., Bethea, C. L. & Roberts, C. T. Jr. Sex-specific differences in lipid and glucose metabolism. Front. Endocrinol. 5, 241 (2014).
Kalyani, R. R. et al. The association of endogenous sex hormones, adiposity, and insulin resistance with incident diabetes in postmenopausal women. J. Clin. Endocrinol. Metab. 94, 4127–4135 (2009).
Google Scholar
Ding, E. L. et al. Plasma sex steroid hormones and risk of developing type 2 diabetes in women: a prospective study. Diabetologia 50, 2076–2084 (2007).
Google Scholar
Karastergiou, K., Smith, S. R., Greenberg, A. S. & Fried, S. K. Sex differences in human adipose tissues — the biology of pear shape. Biol. Sex. Differ. 3, 13 (2012).
Google Scholar
Tchernof, A., Poehlman, E. T. & Despres, J. P. Body fat distribution, the menopause transition, and hormone replacement therapy. Diabetes Metab. 26, 12–20 (2000).
Google Scholar
Palmer, B. F. & Clegg, D. J. The sexual dimorphism of obesity. Mol. Cell Endocrinol. 402, 113–119 (2015).
Google Scholar
Sebo, Z. L. & Rodeheffer, M. S. Testosterone metabolites differentially regulate obesogenesis and fat distribution. Mol. Metab. 44, 101141 (2021).
Google Scholar
Kalkhoff, R. K. Metabolic effects of progesterone. Am. J. Obstet. Gynecol. 142, 735–738 (1982).
Google Scholar
Frank, A. P., de Souza Santos, R., Palmer, B. F. & Clegg, D. J. Determinants of body fat distribution in humans may provide insight about obesity-related health risks. J. Lipid Res. 60, 1710–1719 (2019).
Google Scholar
Cooper, A. J., Gupta, S. R., Moustafa, A. F. & Chao, A. M. Sex/gender differences in obesity prevalence, comorbidities, and treatment. Curr. Obes. Rep. 10, 458–466 (2021).
Google Scholar
Bremer, A. A., Devaraj, S., Afify, A. & Jialal, I. Adipose tissue dysregulation in patients with metabolic syndrome. J. Clin. Endocrinol. Metab. 96, E1782–E1788 (2011).
Google Scholar
Lê, K. A. et al. Subcutaneous adipose tissue macrophage infiltration is associated with hepatic and visceral fat deposition, hyperinsulinemia, and stimulation of NF-κB stress pathway. Diabetes 60, 2802–2809 (2011).
Google Scholar
Carr, M. C. The emergence of the metabolic syndrome with menopause. J. Clin. Endocrinol. Metab. 88, 2404–2411 (2003).
Google Scholar
Bermingham, K. M. et al. Menopause is associated with postprandial metabolism, metabolic health and lifestyle: The ZOE PREDICT study. eBioMedicine 85, 104303 (2022).
Google Scholar
Patel, P. & Abate, N. Body fat distribution and insulin resistance. Nutrients 5, 2019–2027 (2013).
Google Scholar
Susanto, A., Burk, J., Hocking, S., Markovic, T. & Gill, T. Differences in weight loss outcomes for males and females on a low-carbohydrate diet: a systematic review. Obes. Res. Clin. Pract. 16, 447–456 (2022).
Google Scholar
Eaton, S. A. & Sethi, J. K. Immunometabolic links between estrogen, adipose tissue and female reproductive metabolism. Biology 8, 8 (2019).
Google Scholar
Killinger, D. W., Perel, E., Daniilescu, D., Kharlip, L. & Blackstein, M. E. Aromatase activity in the breast and other peripheral tissues and its therapeutic regulation. Steroids 50, 523–536 (1987).
Google Scholar
Bulun, S. E., Chen, D., Moy, I., Brooks, D. C. & Zhao, H. Aromatase, breast cancer and obesity: a complex interaction. Trends Endocrinol. Metab. 23, 83–89 (2012).
Google Scholar
Jones, M. E. et al. Aromatase-deficient (ArKO) mice accumulate excess adipose tissue. J. Steroid Biochem. Mol. Biol. 79, 3–9 (2001).
Google Scholar
Ohlsson, C. et al. Increased adipose tissue aromatase activity improves insulin sensitivity and reduces adipose tissue inflammation in male mice. Am. J. Physiol. Endocrinol. Metab. 313, E450–E462 (2017).
Google Scholar
Steiner, B. M. & Berry, D. C. The regulation of adipose tissue health by estrogens. Front. Endocrinol. 13, 889923 (2022).
Google Scholar
Kozniewski, K. et al. Epigenetic regulation of estrogen receptor genes’ expressions in adipose tissue in the course of obesity. Int. J. Mol. Sci. 23, 5989 (2022).
Google Scholar
Yager, J. D. & Chen, J. Q. Mitochondrial estrogen receptors — new insights into specific functions. Trends Endocrinol. Metab. 18, 89–91 (2007).
Google Scholar
Narayanan, R. et al. Steroidal androgens and nonsteroidal, tissue-selective androgen receptor modulator, S-22, regulate androgen receptor function through distinct genomic and nongenomic signaling pathways. Mol. Endocrinol. 22, 2448–2465 (2008).
Google Scholar
Sharma, G., Mauvais-Jarvis, F. & Prossnitz, E. R. Roles of G protein-coupled estrogen receptor GPER in metabolic regulation. J. Steroid Biochem. Mol. Biol. 176, 31–37 (2018).
Google Scholar
Haas, E. et al. Regulatory role of G protein-coupled estrogen receptor for vascular function and obesity. Circ. Res. 104, 288–291 (2009).
Google Scholar
Davis, K. E. et al. Sexually dimorphic role of G protein-coupled estrogen receptor (GPER) in modulating energy homeostasis. Horm. Behav. 66, 196–207 (2014).
Google Scholar
Sharma, G. & Prossnitz, E. R. Targeting the G protein-coupled estrogen receptor (GPER) in obesity and diabetes. Endocr. Metab. Sci. 2, 100080 (2021).
Google Scholar
Prossnitz, E. R. & Barton, M. The G protein-coupled oestrogen receptor GPER in health and disease: an update. Nat. Rev. Endocrinol. 19, 407–424 (2023).
Google Scholar
Roepke, T. A., Ronnekleiv, O. K. & Kelly, M. J. Physiological consequences of membrane-initiated estrogen signaling in the brain. Front. Biosci. 16, 1560–1573 (2011).
Google Scholar
Toran-Allerand, C. D. et al. ER-X: a novel, plasma membrane-associated, putative estrogen receptor that is regulated during development and after ischemic brain injury. J. Neurosci. 22, 8391–8401 (2002).
Google Scholar
Tang, Z. R., Zhang, R., Lian, Z. X., Deng, S. L. & Yu, K. Estrogen-receptor expression and function in female reproductive disease. Cells 8, 1123 (2019).
Google Scholar
Luo, F. et al. 17β-estradiol lowers triglycerides in adipocytes via estrogen receptor α and it may be attenuated by inflammation. Lipids Health Dis. 16, 182 (2017).
Google Scholar
Yepuru, M. et al. Estrogen receptor-β-selective ligands alleviate high-fat diet- and ovariectomy-induced obesity in mice. J. Biol. Chem. 285, 31292–31303 (2010).
Google Scholar
Heine, P. A., Taylor, J. A., Iwamoto, G. A., Lubahn, D. B. & Cooke, P. S. Increased adipose tissue in male and female estrogen receptor-α knockout mice. Proc. Natl Acad. Sci. USA 97, 12729–12734 (2000).
Google Scholar
Cooke, P. S., Heine, P. A., Taylor, J. A. & Lubahn, D. B. The role of estrogen and estrogen receptor-α in male adipose tissue. Mol. Cell Endocrinol. 178, 147–154 (2001).
Google Scholar
Winn, N. C. et al. Estrogen receptor-α signaling maintains immunometabolic function in males and is obligatory for exercise-induced amelioration of nonalcoholic fatty liver. Am. J. Physiol. Endocrinol. Metab. 316, E156–E167 (2019).
Google Scholar
Ponnusamy, S. et al. Pharmacologic activation of estrogen receptor β increases mitochondrial function, energy expenditure, and brown adipose tissue. FASEB J. 31, 266–281 (2017).
Google Scholar
Clegg, D. et al. Sex hormones and cardiometabolic health: role of estrogen and estrogen receptors. Endocrinology 158, 1095–1105 (2017).
Google Scholar
Davis, K. E. et al. The sexually dimorphic role of adipose and adipocyte estrogen receptors in modulating adipose tissue expansion, inflammation, and fibrosis. Mol. Metab. 2, 227–242 (2013).
Google Scholar
Weigt, C., Hertrampf, T., Zoth, N., Fritzemeier, K. H. & Diel, P. Impact of estradiol, ER subtype specific agonists and genistein on energy homeostasis in a rat model of nutrition induced obesity. Mol. Cell Endocrinol. 351, 227–238 (2012).
Google Scholar
Misso, M. L. et al. Cellular and molecular characterization of the adipose phenotype of the aromatase-deficient mouse. Endocrinology 144, 1474–1480 (2003).
Google Scholar
Cowley, S. M., Hoare, S., Mosselman, S. & Parker, M. G. Estrogen receptors alpha and beta form heterodimers on DNA. J. Biol. Chem. 272, 19858–19862 (1997).
Google Scholar
Ahmed, F. et al. Role of estrogen and its receptors in adipose tissue glucose metabolism in pre- and postmenopausal women. J. Clin. Endocrinol. Metab. 107, e1879–e1889 (2022).
Google Scholar
Porter, J. W. et al. Age, sex, and depot-specific differences in adipose-tissue estrogen receptors in individuals with obes. Obesity 28, 1698–1707 (2020).
Google Scholar
Zidon, T. M. et al. Effects of ERβ and ERα on OVX-induced changes in adiposity and insulin resistance. J. Endocrinol. 245, 165–178 (2020).
Google Scholar
Foryst-Ludwig, A. et al. Metabolic actions of estrogen receptor beta (ERβ) are mediated by a negative cross-talk with PPARγ. PLoS Genet. 4, e1000108 (2008).
Google Scholar
Lefterova, M. I., Haakonsson, A. K., Lazar, M. A. & Mandrup, S. PPARγ and the global map of adipogenesis and beyond. Trends Endocrinol. Metab. 25, 293–302 (2014).
Google Scholar
Iorga, A. et al. The protective role of estrogen and estrogen receptors in cardiovascular disease and the controversial use of estrogen therapy. Biol. Sex. Differ. 8, 33 (2017).
Google Scholar
Gerdts, E. & Regitz-Zagrosek, V. Sex differences in cardiometabolic disorders. Nat. Med. 25, 1657–1666 (2019).
Google Scholar
Norhammar, A. Diabetes and cardiovascular mortality: the impact of sex. Lancet Diabetes Endocrinol. 6, 517–519 (2018).
Google Scholar
Martinez-Cignoni, M. R. et al. Estrogen impairs adipose tissue expansion and cardiometabolic profile in obese-diabetic female rats. Int. J. Mol. Sci. 22, 13573 (2021).
Google Scholar
Chattopadhyay, S. et al. Estradiol overcomes adiponectin-resistance in diabetic mice by regulating skeletal muscle adiponectin receptor 1 expression. Mol. Cell Endocrinol. 540, 111525 (2022).
Google Scholar
Sieminska, L. et al. Effect of postmenopause and hormone replacement therapy on serum adiponectin levels. Metabolism 54, 1610–1614 (2005).
Google Scholar
Tchkonia, T. et al. Mechanisms and metabolic implications of regional differences among fat depots. Cell Metab. 17, 644–656 (2013).
Google Scholar
Panarotto, D., Remillard, P., Bouffard, L. & Maheux, P. Insulin resistance affects the regulation of lipoprotein lipase in the postprandial period and in an adipose tissue-specific manner. Eur. J. Clin. Invest. 32, 84–92 (2002).
Google Scholar
Chait, A. & den Hartigh, L. J. Adipose tissue distribution, inflammation and its metabolic consequences, including diabetes and cardiovascular disease. Front. Cardiovasc. Med. 7, 22 (2020).
Google Scholar
Meyer, L. K., Ciaraldi, T. P., Henry, R. R., Wittgrove, A. C. & Phillips, S. A. Adipose tissue depot and cell size dependency of adiponectin synthesis and secretion in human obesity. Adipocyte 2, 217–226 (2013).
Google Scholar
Park, Y. M. et al. Estradiol-mediated improvements in adipose tissue insulin sensitivity are related to the balance of adipose tissue estrogen receptor alpha and beta in postmenopausal women. PLoS ONE 12, e0176446 (2017).
Google Scholar
Kurylowicz, A. Estrogens in adipose tissue physiology and obesity-related dysfunction. Biomedicines 11, 690 (2023).
Google Scholar
Vieira Potter, V. J. et al. Adipose tissue inflammation and reduced insulin sensitivity in ovariectomized mice occurs in the absence of increased adiposity. Endocrinology 153, 4266–4277 (2012).
Google Scholar
Fisher, C. R., Graves, K. H., Parlow, A. F. & Simpson, E. R. Characterization of mice deficient in aromatase (ArKO) because of targeted disruption of the cyp19 gene. Proc. Natl Acad. Sci. USA 95, 6965–6970 (1998).
Google Scholar
Shay, D. A. et al. Changes in nucleus accumbens gene expression accompany sex-specific suppression of spontaneous physical activity in aromatase knockout mice. Horm. Behav. 121, 104719 (2020).
Google Scholar
Harding, A. T. & Heaton, N. S. The impact of estrogens and their receptors on immunity and inflammation during infection. Cancers 14, 909 (2022).
Google Scholar
Harding, A. T., Goff, M. A., Froggatt, H. M., Lim, J. K. & Heaton, N. S. GPER1 is required to protect fetal health from maternal inflammation. Science 371, 271–276 (2021).
Google Scholar
Vieira-Potter, V. J. Inflammation and macrophage modulation in adipose tissues. Cell Microbiol. 16, 1484–1492 (2014).
Google Scholar
Xie, X. et al. Role of adipocyte mitochondria in inflammation, lipemia and insulin sensitivity in humans: effects of pioglitazone treatment. Int. J. Obes. 42, 213–220 (2017).
Google Scholar
Heinonen, S. et al. Mitochondria-related transcriptional signature is downregulated in adipocytes in obesity: a study of young healthy MZ twins. Diabetologia 60, 169–181 (2017).
Google Scholar
Capllonch-Amer, G., Llado, I., Proenza, A. M., Garcia-Palmer, F. J. & Gianotti, M. Opposite effects of 17-beta estradiol and testosterone on mitochondrial biogenesis and adiponectin synthesis in white adipocytes. J. Mol. Endocrinol. 52, 203–214 (2014).
Google Scholar
Kim, S. N. et al. Sex differences in sympathetic innervation and browning of white adipose tissue of mice. Biol. Sex. Differ. 7, 67 (2016).
Google Scholar
Pedersen, S. B., Bruun, J. M., Kristensen, K. & Richelsen, B. Regulation of UCP1, UCP2, and UCP3 mRNA expression in brown adipose tissue, white adipose tissue, and skeletal muscle in rats by estrogen. Biochem. Biophys. Res. Commun. 288, 191–197 (2001).
Google Scholar
Nookaew, I. et al. Adipose tissue resting energy expenditure and expression of genes involved in mitochondrial function are higher in women than in men. J. Clin. Endocrinol. Metab. 98, E370–E378 (2013).
Google Scholar
Asterholm, I. W. & Scherer, P. E. Enhanced metabolic flexibility associated with elevated adiponectin levels. Am. J. Pathol. 176, 1364–1376 (2010).
Google Scholar
Sanchez, M. I. et al. Estrogen-mediated regulation of mitochondrial gene expression. Mol. Endocrinol. 29, 14–27 (2015).
Google Scholar
Zhou, Z. et al. Estrogen receptor α controls metabolism in white and brown adipocytes by regulating Polg1 and mitochondrial remodeling. Sci. Transl. Med. 12, eaax8096 (2020).
Google Scholar
Simpkins, J. W., Yang, S. H., Sarkar, S. N. & Pearce, V. Estrogen actions on mitochondria-physiological and pathological implications. Mol. Cell Endocrinol. 290, 51–59 (2008).
Google Scholar
Hamilton, D. J. et al. Estrogen receptor alpha activation enhances mitochondrial function and systemic metabolism in high-fat-fed ovariectomized mice. Physiol. Rep. 4, e12913 (2016).
Google Scholar
Sasayama, D. et al. Novel oestrogen receptor β-selective ligand reduces obesity and depressive-like behaviour in ovariectomized mice. Sci. Rep. 7, 4663 (2017).
Google Scholar
Gonzalez-Granillo, M. et al. ERβ activation in obesity improves whole body metabolism via adipose tissue function and enhanced mitochondria biogenesis. Mol. Cell Endocrinol. 479, 147–158 (2019).
Google Scholar
Song, I. S. et al. Modulation of mitochondrial ERβ expression inhibits triple-negative breast cancer tumor progression by activating mitochondrial function. Cell Physiol. Biochem. 52, 468–485 (2019).
Google Scholar
Yang, S. H. et al. Mitochondrial localization of estrogen receptor β. Proc. Natl Acad. Sci. USA 101, 4130–4135 (2004).
Google Scholar
Chen, J. Q., Eshete, M., Alworth, W. L. & Yager, J. D. Binding of MCF-7 cell mitochondrial proteins and recombinant human estrogen receptors α and β to human mitochondrial DNA estrogen response elements. J. Cell Biochem. 93, 358–373 (2004).
Google Scholar
Himms-Hagen, J. Brown adipose tissue thermogenesis and obesity. Prog. Lipid Res. 28, 67–115 (1989).
Google Scholar
Cypess, A. M. et al. Identification and importance of brown adipose tissue in adult humans. N. Engl. J. Med. 360, 1509–1517 (2009).
Google Scholar
Quevedo, S., Roca, P., Pico, C. & Palou, A. Sex-associated differences in cold-induced UCP1 synthesis in rodent brown adipose tissue. Eur. J. Physiol. 436, 689–695 (1998).
Google Scholar
Townsend, K. L. & Tseng, Y. H. Brown fat fuel utilization and thermogenesis. Trends Endocrinol. Metab. 25, 168–177 (2014).
Google Scholar
Clookey, S. L. et al. Beta 3 adrenergic receptor activation rescues metabolic dysfunction in female estrogen receptor alpha-null mice. Front. Physiol. 10, 9 (2019).
Google Scholar
Bloom, J. D. et al. Disodium (R,R)-5-[2-[[2-(3-chlorophenyl)-2-hydroxyethyl]-amino] propyl]-1,3-benzodioxole-2,2-dicarboxylate (CL 316,243). A potent β-adrenergic agonist virtually specific for β3 receptors. A promising antidiabetic and antiobesity agent. J. Med. Chem. 35, 3081–3084 (1992).
Google Scholar
Himms-Hagen, J. et al. Effect of CL-316,243, a thermogenic beta 3-agonist, on energy balance and brown and white adipose tissues in rats. Am. J. Physiol. 266, R1371–R1382 (1994).
Google Scholar
Grujic, D. et al. β3-adrenergic receptors on white and brown adipocytes mediate β3-selective agonist-induced effects on energy expenditure, insulin secretion, and food intake. A study using transgenic and gene knockout mice. J. Biol. Chem. 272, 17686–17693 (1997).
Google Scholar
Blondin, D. P. et al. Brown adipose tissue metabolism in women is dependent on ovarian status. Am. J. Physiol. Endocrinol. Metab. 326, E588–E601 (2024).
Google Scholar
Yu, Z., Jiao, Y., Zhao, Y. & Gu, W. Level of estrogen in females — the different impacts at different life stages. J. Pers. Med. 12, 1995 (2022).
Google Scholar
Petrakis, N. L. et al. Influence of pregnancy and lactation on serum and breast fluid estrogen levels: implications for breast cancer risk. Int. J. Cancer 40, 587–591 (1987).
Google Scholar
Amoah, C., Adageba, R. K., Appiah, E. K. & Sefogah, P. E. Obesity and overweight and associated factors among women with infertility undergoing assisted reproductive technology treatment in a low income setting. Sci. Rep. 15, 6163 (2025).
Google Scholar
Yildiz, B. O. & Azziz, R. Ovarian and adipose tissue dysfunction in polycystic ovary syndrome: report of the 4th Special Scientific Meeting of the Androgen Excess and PCOS Society. Fertil. Steril. 94, 690–693 (2010).
Google Scholar
Maloney, A. C., Barnas, J. L., Clart, L. M., Vieira-Potter, V. J. & Kanaley, J. A. Metabolic dysfunction is exacerbated in visceral, not subcutaneous, adipose tissue in gestational diabetes. J. Endocrinol. 265, e250043 (2025).
Google Scholar
Trivett, C., Lees, Z. J. & Freeman, D. J. Adipose tissue function in healthy pregnancy, gestational diabetes mellitus and pre-eclampsia. Eur. J. Clin. Nutr. 75, 1745–1756 (2021).
Google Scholar
Subramanian, N. et al. Adipokine secretion and lipolysis following gender-affirming treatment in transgender individuals. J. Endocrinol. Invest. 47, 2249–2260 (2024).
Google Scholar
Hetemäki, N. et al. Adipose tissue estrogen production and metabolism in premenopausal women. J. Steroid Biochem. Mol. Biol. 209, 105849 (2021).
Google Scholar
Yeung, E. H. et al. Adiposity and sex hormones across the menstrual cycle: the BioCycle Study. Int. J. Obes. 37, 237–243 (2013).
Google Scholar
MacGregor, K. A. et al. Changes in adipose tissue microRNA expression across the menstrual cycle in regularly menstruating females: a pilot study. Physiol. Genom. 54, 1–10 (2022).
Google Scholar
Marsden, P. J., Murdoch, A. & Taylor, R. Adipocyte insulin action during the normal menstrual cycle. Hum. Reprod. 11, 968–974 (1996).
Google Scholar
Rebuffé-Scrive, M. et al. Fat cell metabolism in different regions in women. Effect of menstrual cycle, pregnancy, and lactation. J. Clin. Invest. 75, 1973–1976 (1985).
Google Scholar
Goulart, A. C., Zee, R. Y. & Rexrode, K. M. Estrogen receptor 1 gene polymorphisms and decreased risk of obesity in women. Metabolism 58, 759–764 (2009).
Google Scholar
Nilsson, M. et al. Impact of estrogen receptor gene polymorphisms and mRNA levels on obesity and lipolysis — a cohort study. BMC Med. Genet. 8, 73 (2007).
Google Scholar
Shin, J. H. et al. The ratio of estrogen receptor α to estrogen receptor β in adipose tissue is associated with leptin production and obesity. Steroids 72, 592–599 (2007).
Google Scholar
Kersten, S. Peroxisome proliferator activated receptors and lipoprotein metabolism. PPAR Res. 2008, 132960 (2008).
Google Scholar
Hirschenson, J., Melgar-Bermudez, E. & Mailloux, R. J. The uncoupling proteins: a systematic review on the mechanism used in the prevention of oxidative stress. Antioxidants 11, 322 (2022).
Google Scholar
Shen, K. et al. Anticancer or carcinogenic? The role of estrogen receptor β in breast cancer progression. Pharmacol. Ther. 242, 108350 (2023).
Google Scholar
Cleary, M. P. & Grossmann, M. E. Minireview: obesity and breast cancer: the estrogen connection. Endocrinology 150, 2537–2542 (2009).
Google Scholar
Turek, J. & Gasior, L. Estrogen fluctuations during the menopausal transition are a risk factor for depressive disorders. Pharmacol. Rep. 75, 32–43 (2023).
Google Scholar
Frank, A., Brown, L. M. & Clegg, D. J. The role of hypothalamic estrogen receptors in metabolic regulation. Front. Neuroendocrinol. 35, 550–557 (2014).
Google Scholar
Martinez de Morentin, P. B. et al. Estradiol regulates brown adipose tissue thermogenesis via hypothalamic AMPK. Cell Metab. 20, 41–53 (2014).
Google Scholar
Maioli, S., Leander, K., Nilsson, P. & Nalvarte, I. Estrogen receptors and the aging brain. Essays Biochem. 65, 913–925 (2021).
Google Scholar
van Veen, J. E. et al. Hypothalamic estrogen receptor alpha establishes a sexually dimorphic regulatory node of energy expenditure. Nat. Metab. 2, 351–363 (2020).
Google Scholar
McEwen, B. Estrogen actions throughout the brain. Recent. Prog. Horm. Res. 57, 357–384 (2002).
Google Scholar
Xu, Y. et al. Distinct hypothalamic neurons mediate estrogenic effects on energy homeostasis and reproduction. Cell Metab. 14, 453–465 (2011).
Google Scholar
Wang, A. et al. GPR30 regulates diet-induced adiposity in female mice and adipogenesis in vitro. Sci. Rep. 6, 34302 (2016).
Google Scholar
Almey, A., Milner, T. A. & Brake, W. G. Estrogen receptors observed at extranuclear neuronal sites and in glia in the nucleus accumbens core and shell of the female rat: evidence for localization to catecholaminergic and GABAergic neurons. J. Comp. Neurol. 530, 2056–2072 (2022).
Google Scholar
Shay, D. et al. Knockdown of Esr1 from DRD1-rich brain regions affects adipose tissue metabolism: potential crosstalk between nucleus accumbens and adipose tissue. Int. J. Mol. Sci. 25, 6130 (2024).
Google Scholar
Gavin, K. M., Kohrt, W. M., Klemm, D. J. & Melanson, E. L. Modulation of energy expenditure by estrogens and exercise in women. Exerc. Sport. Sci. Rev. 46, 232–239 (2018).
Google Scholar
Musatov, S. et al. Silencing of estrogen receptor α in the ventromedial nucleus of hypothalamus leads to metabolic syndrome. Proc. Natl Acad. Sci. USA 104, 2501–2506 (2007).
Google Scholar
Vacca, V. et al. 17β-estradiol counteracts neuropathic pain: a behavioural, immunohistochemical, and proteomic investigation on sex-related differences in mice. Sci. Rep. 6, 18980 (2016).
Google Scholar
Deng, C., Gu, Y. J., Zhang, H. & Zhang, J. Estrogen affects neuropathic pain through upregulating N-methyl-D-aspartate acid receptor 1 expression in the dorsal root ganglion of rats. Neural Regen. Res. 12, 464–469 (2017).
Google Scholar
Singh, A., Asif, N., Singh, P. N. & Hossain, M. M. Motor nerve conduction velocity in postmenopausal women with peripheral neuropathy. J. Clin. Diagn. Res. 10, CC13–CC16 (2016).
Google Scholar
Chen, Q., Zhang, W., Sadana, N. & Chen, X. Estrogen receptors in pain modulation: cellular signaling. Biol. Sex. Differ. 12, 22 (2021).
Google Scholar
Chen, Y. et al. 17β-estradiol promotes Schwann cell proliferation and differentiation, accelerating early remyelination in a mouse peripheral nerve injury model. Biomed. Res. Int. 2016, 7891202 (2016).
Google Scholar
Patrone, C., Andersson, S., Korhonen, L. & Lindholm, D. Estrogen receptor-dependent regulation of sensory neuron survival in developing dorsal root ganglion. Proc. Natl Acad. Sci. USA 96, 10905–10910 (1999).
Google Scholar
Toran-Allerand, C. D., Tinnikov, A. A., Singh, R. J. & Nethrapalli, I. S. 17α-estradiol: a brain-active estrogen? Endocrinology 146, 3843–3850 (2005).
Google Scholar
Stout, M. B. et al. 17α-estradiol alleviates age-related metabolic and inflammatory dysfunction in male mice without inducing feminization. J. Gerontol. A Biol. Sci. Med. Sci. 72, 3–15 (2017).
Google Scholar
Mann, S. N. et al. Health benefits attributed to 17α-estradiol, a lifespan-extending compound, are mediated through estrogen receptor α. eLife 9, e59616 (2020).
Google Scholar
Garratt, M. et al. Male lifespan extension with 17-α estradiol is linked to a sex-specific metabolomic response modulated by gonadal hormones in mice. Aging Cell 17, e12786 (2018).
Google Scholar
Vieira-Potter, V. J. et al. Female rats selectively bred for high intrinsic aerobic fitness are protected from ovariectomy-associated metabolic dysfunction. Am. J. Physiol. Regul. Integr. Comp. Physiol. 308, R530–R542 (2015).
Google Scholar
Welly, R. J. et al. Comparison of diet versus exercise on metabolic function and gut microbiota in obese rats. Med. Sci. Sports Exerc. 48, 1688–1698 (2016).
Google Scholar
Zidon, T. M. et al. Voluntary wheel running improves adipose tissue immunometabolism in ovariectomized low-fit rats. Adipocyte 7, 20–34 (2018).
Google Scholar
Pallottini, V., Bulzomi, P., Galluzzo, P., Martini, C. & Marino, M. Estrogen regulation of adipose tissue functions: involvement of estrogen receptor isoforms. Infect. Disord. Drug. Targets 8, 52–60 (2008).
Google Scholar
Vieira-Potter, V. J., Zidon, T. M. & Padilla, J. Exercise and estrogen make fat cells “fit”. Exerc. Sport. Sci. Rev. 43, 172–178 (2015).
Google Scholar
Marsh, M. L., Oliveira, M. N. & Vieira-Potter, V. J. Adipocyte metabolism and health after the menopause: the role of exercise. Nutrients 15, 444 (2023).
Google Scholar
Park, Y. M. et al. Voluntary running attenuates metabolic dysfunction in ovariectomized low-fit rats. Med. Sci. Sports Exerc. 49, 254–264 (2017).
Google Scholar
Park, Y. M. et al. Effects of ovariectomy and intrinsic aerobic capacity on tissue-specific insulin sensitivity. Am. J. Physiol. Endocrinol. Metab. 310, E190–E199 (2016).
Google Scholar
Dam, T. V. et al. Estrogen modulates metabolic risk profile after resistance training in early postmenopausal women: a randomized controlled trial. Menopause 28, 1214–1224 (2021).
Google Scholar
Dam, T. V. et al. Transdermal estrogen therapy improves gains in skeletal muscle mass after 12 weeks of resistance training in early postmenopausal women. Front. Physiol. 11, 596130 (2020).
Google Scholar
Kim, D. I. & Park, Y. M. Effects of menopause on physical activity and dopamine signaling in women. Iran. J. Public. Health 51, 2372–2374 (2022).
Google Scholar
Razmjou, S. et al. Effect of the menopausal transition and physical activity energy expenditure on inflammatory markers: a MONET group study. Menopause 23, 1330–1338 (2016).
Google Scholar
Park, Y. M. et al. Effects of intrinsic aerobic capacity and ovariectomy on voluntary wheel running and nucleus accumbens dopamine receptor gene expression. Physiol. Behav. 164, 383–389 (2016).
Google Scholar
Cleveland Clinic. Selective estrogen receptor modulators (SERMs). Cleveland Clinic (2023).
Manson, J. E. et al. Menopausal hormone therapy and health outcomes during the intervention and extended poststopping phases of the Women’s Health Initiative randomized trials. JAMA 310, 1353–1368 (2013).
Google Scholar
Hodis, H. N. & Mack, W. J. Menopausal hormone replacement therapy and reduction of all-cause mortality and cardiovascular disease: it is about time and timing. Cancer J. 28, 208–223 (2022).
Google Scholar
El Khoudary, S. R. et al. Menopause transition and cardiovascular disease risk: implications for timing of early prevention: a scientific statement from the American Heart Association. Circulation 142, e506–e532 (2020).
Google Scholar
Iyengar, N. M., Hudis, C. A. & Dannenberg, A. J. Obesity and inflammation: new insights into breast cancer development and progression. Am. Soc. Clin. Oncol. Educ. Book. 33, 46–51 (2013).
Google Scholar
Yi, K. D. et al. The assessment of non-feminizing estrogens for use in neuroprotection. Brain Res. 1379, 61–70 (2011).
Google Scholar
Gonzalez-Granillo, M. et al. Selective estrogen receptor (ER)β activation provokes a redistribution of fat mass and modifies hepatic triglyceride composition in obese male mice. Mol. Cell Endocrinol. 502, 110672 (2020).
Google Scholar
Queathem, E. D. et al. Suppression of estrogen receptor beta classical genomic activity enhances systemic and adipose-specific response to chronic beta-3 adrenergic receptor (β3AR) stimulation. Front. Physiol. 13, 920675 (2022).
Google Scholar
Sedlak, D. et al. Structure–activity relationship of para-carborane selective estrogen receptor β agonists. J. Med. Chem. 64, 9330–9353 (2021).
Google Scholar
Park, Y. M., Myers, M. & Vieira-Potter, V. J. Adipose tissue inflammation and metabolic dysfunction: role of exercise. Mo. Med. 111, 65–72 (2014).
Google Scholar
Malandish, A. & Rahmati-Yamchi, M. The effect of moderate intensity aerobic exercise on cardiovascular function, cardiorespiratory fitness and estrogen receptor alpha gene in overweight/obese postmenopausal women: a randomized controlled trial. J. Mol. Cell. Cardiol. 2, 100026 (2022).
Haines, M. et al. Skeletal muscle estrogen receptor activation in response to eccentric exercise up-regulates myogenic-related gene expression independent of differing serum estradiol levels occurring during the human menstrual cycle. J. Sports Sci. Med. 17, 31–39 (2018).
Google Scholar
Liu, Y., Nguyen, N. & Colditz, G. A. Links between alcohol consumption and breast cancer: a look at the evidence. Women’s Health 11, 65–77 (2015).
Google Scholar
Justenhoven, C., Obazee, O. & Brauch, H. The pharmacogenomics of sex hormone metabolism: breast cancer risk in menopausal hormone therapy. Pharmacogenomics 13, 659–675 (2012).
Google Scholar
Abou-Ismail, M. Y., Citla Sridhar, D. & Nayak, L. Estrogen and thrombosis: a bench to bedside review. Thromb. Res. 192, 40–51 (2020).
Google Scholar
Cho, L. et al. Rethinking menopausal hormone therapy: for whom, what, when, and how long? Circulation 147, 597–610 (2023).
Google Scholar
Samaras, K., Hayward, C. S., Sullivan, D., Kelly, R. P. & Campbell, L. V. Effects of postmenopausal hormone replacement therapy on central abdominal fat, glycemic control, lipid metabolism, and vascular factors in type 2 diabetes: a prospective study. Diabetes Care 22, 1401–1407 (1999).
Google Scholar
Kleis-Olsen, A. S. et al. Metabolic flexibility in postmenopausal women: hormone replacement therapy is associated with higher mitochondrial content, respiratory capacity, and lower total fat mass. Acta Physiol. 240, e14117 (2024).
Google Scholar
Javed, A. A., Mayhew, A. J., Shea, A. K. & Raina, P. Association between hormone therapy and muscle mass in postmenopausal women: a systematic review and meta-analysis. JAMA Netw. Open. 2, e1910154 (2019).
Google Scholar
Liu, S. H. et al. Estrogen restores disordered lipid metabolism in visceral fat of prediabetic mice. World J. Diabetes 15, 988–1000 (2024).
Google Scholar
Kalenga, C. Z. et al. Association between the route of administration and formulation of estrogen therapy and hypertension risk in postmenopausal women: a prospective population-based study. Hypertension 80, 1463–1473 (2023).
Google Scholar
El-Tanani, M. K. & Green, C. D. Two separate mechanisms for ligand-independent activation of the estrogen receptor. Mol. Endocrinol. 11, 928–937 (1997).
Google Scholar
Carlson, K. E., Choi, I., Gee, A., Katzenellenbogen, B. S. & Katzenellenbogen, J. A. Altered ligand binding properties and enhanced stability of a constitutively active estrogen receptor: evidence that an open pocket conformation is required for ligand interaction. Biochemistry 36, 14897–14905 (1997).
Google Scholar
link